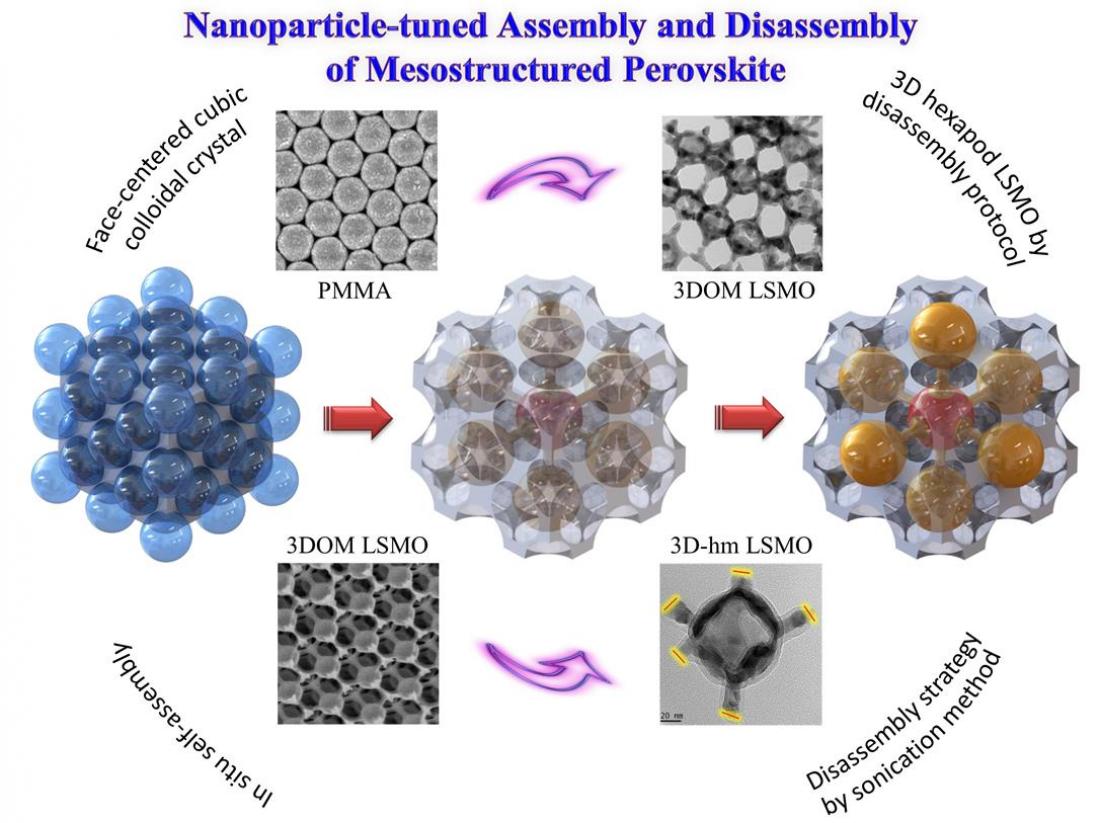
Schematic illustration depicting synthesis of the 3D-hm LSMO catalysts.
Versatile superstructures composed of nanoparticles have recently been prepared using various disassembly methods. However, little information is known on how the structural disassembly influences the catalytic performance of the materials. Scientia Professor Rose Amal, Vice-Chancellor’s Research Fellow Hamid Arandiyan and a group from the Particles and Catalysis Research Group from the University of New South Wales (UNSW) School of Chemical Engineering have had their research address this issue published in Nature Communications.
The research team led by Dr Jason Scott and Prof Sean Smith in collaboration with Curtin University and Beijing University of Technology has developed a method that allows them to engineer crystals with a large fraction of reactive facets. An ordered mesostructured La0.6Sr0.4MnO3 (LSMO) perovskite catalyst was disassembled using a unique fragmentation strategy, whereby the newly-exposed (001) reactive faces at each fracture were more reactive towards methane oxidation than the regular (i.e. before disassembly)
It is of significant interest to use methane as an alternative fuel to coal and oil due to its high hydrogen to carbon ratio which provides comparatively lower greenhouse gas emissions. Commercial catalysts for methane combustion contain precious metals (e.g. Pt and Pd) which are of high cost and poor thermal stability (caused by agglomeration of the metal deposits). Using perovskite-type catalysts to replace noble metal supported catalysts for methane oxidation has attracted recent attention due to their excellent thermal stability. In their recently published article, the research team describes a simple fragmentation method to synthesise a novel three-dimensional hexapod mesostructured LSMO perovskite.
On fragmenting three-dimensionally ordered macroporous (3DOM) structures in a controlled manner, via a process that has been likened to retrosynthesis, hexapod-shaped building blocks possessing newly exposed active crystal facets were harvested. Powerful characteristation techniques were coupled with theoretical calculations to define the manner by which the improved configuration promotes the methane combustion reaction.
The new (110) reactive facets exposed at the weak fracture points of the 3DOM structure provide additional surface area as well as introduce surfaces possessing a reduced energy barrier for hydrogen abstraction from the methane (CH4* → CH3* + H*) compared to the regular 3DOM (001) nonreactive facets. We believe the design philosophy and the preparation strategy for 3D LSMO provides an original pathway towards engineering high-efficiency catalysts.
The fragmentation technique can be extended to the controlled preparation and stabilization of other nanomaterials with broad applications, for this reason, it is of great significance. The approach demonstrates feasibility, “mesoporous material field is eager for more and more researchers from other fields to explore attractive applications,” says PhD student Yuan Wang from Particle and Catalysis (PartCat) Research Group. “there is still ample room for improvement on hierarchically ordered perovskite catalysts designed to reduce atmospheric greenhouse gas concentrations by oxidising methane emissions and therefore improve cost-effectiveness,” adds Dr. Hamid Arandiyan of PartCat Research Group.
For more information please contact
Dr. Hamid Arandiyan
UNSW Vice-Chancellor's Research Fellow
School of Chemical Engineering
Particles & Catalysis Research Group