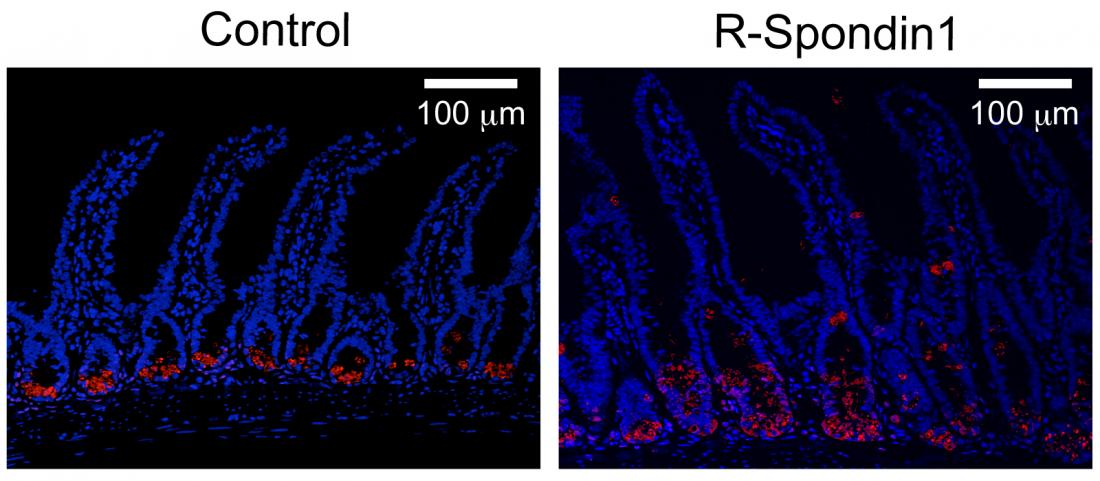
The administration of R -Spondin1 resulted in the increased secretion of α-defensin (right) compared to the control experiment (left).
The trillions of microbes living in a mammal’s intestine play an important role in the host’s metabolism and immunity. The composition of microbiota is maintained by antimicrobial proteins secreted from intestinal cells. However, unfavorably altered microbiota, also known as dysbiosis, is seen in various diseases often exacerbating the underlying disease and thus creating a vicious cycle between the host and the microbiota.
In order to restore microbiota to a normal state, “bacteriotherapy” such as fecal microbiota transplantation and the use of probiotics has been implemented, but no physiological approach has yet been developed so far.
In experiments using mice, a team of scientists led by Professor Takanori Teshima of Hokkaido University discovered that the molecule R-Spondin1 stimulates intestinal stem cells to differentiate to Paneth cells which secrete antimicrobial peptides. The peptides are called α-defensins and are known to have strong and selective antimicrobial activities against pathological bacteria.
When they administered R-Spondin1 into healthy mice, they detected a remarkable increase in the number of Paneth cells and amount of α-defensin secretion while no significant changes in gut microbiota was found. This indicated that α-defensin does not kill symbiotic microbes in a healthy gut.
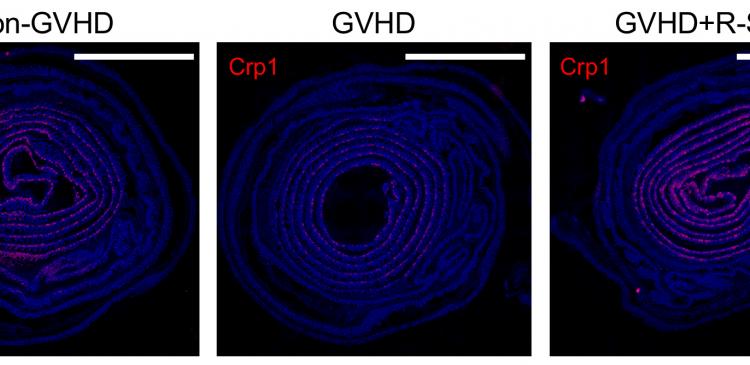
The team then used mice with graft-versus-host-disease (GVHD), an inflammatory complication that occurs after allogenic hematopoietic stem cell transplantation. The team has previously shown that GVHD leads to the loss of Paneth cells and induces intestinal dysbiosis. They have now found that the administration of R-Spondin1 prevents the GVHD-mediated depletion of Paneth cells and the decreased secretion of α-defensins. Importantly, the composition of intestinal microbiota was largely restored by the administration of R-Spondin1. The team also found evidence that α-defensin has similar effects on GVHD-mediated abnormalities when orally administered.
“The two molecules we tested, R-Spondin1 and α-defensin, are both intrinsic to mammals, including humans, and found to have little effect on healthy microbiota. So, our results suggest a novel and physiological approach to restore the gut’s ecosystem and homeostasis while avoiding adverse effects,” says Takanori Teshima.
Contacts:
Professor Takanori Teshima
Department of Hematology
Graduate School of Medicine
Hokkaido University
Email: teshima[at]med.hokudai.ac.jp
Naoki Namba (Media Officer)
Global Relations Office
Institute for International Collaboration
Hokkaido University
Tel: +81-11-706-2185
Email: pr[at]oia.hokudai.ac.jp