A research group led by Takashi Tsuji (Professor in the Research Institute for Science and Technology, Tokyo University of Science, and Director of Organ Technologies Inc.) has demonstrated in growing new organs in adult mice. Tsuji is a research team member in “Health Labor Sciences Research Grant: Research on Regenerative Medicine for Clinical Application (Domain Leader: Professor Akira Yamaguchi of Tokyo Medical and Dental University)”, and “Priority Domain Research: Bio-engineering (Domain Leader: Professor Toshio Fukuda of Nagoya University)”. In transplantation experiments using the tooth as a model, a bioengineered tooth germ develops into a fully functioning bioengineered tooth with sufficient hardness for mastication and a functional responsiveness to mechanical stress in the maxillofacial region. The research also provided the results that the nerve fibers that have re-entered the pulp and periodontal ligament (PDL) tissues of the bioengineered tooth have proper perceptive potential in response to noxious stimulations such as orthodontic treatment and pulp stimulation.
This research is expected to substantially advance in the development of “tooth regenerative therapy”, which have potential as next-generation regenerative therapies for replacing diseased or damaged teeth with bioengineered teeth. Specifically it will not only promote “tooth regenerative therapy”, whereby organ germs of bioengineered teeth are transplanted into the jaw bone to grow “3rd generation tooth”, but is expected to evolve into a wide variety of organ regenerative technologies for liver, kidney and other organs.
This research outcome was the fruit of joint research with Professor Teruko Takano-Yamamoto (Division of Orthodontics and Dentofacial Orthopedics, Graduate School of Dentistry, Tohoku University, Japan) and Professor Shohei Kasugai (Oral and Maxillofacial Surgery, Department of Oral Restitution, Division of Oral Health Sciences, Graduate School, Tokyo Medical and Dental University, Japan). It was announced in an Advance Online Publication of the US scientific journal “Proc. Natl. Acad. Sci. USA” at 17:00hrs (Eastern US Time) on Aug. 03, 2009.
Background to the research
Regenerative therapy is expected to be one of the novel clinical systems in the 21st century. One of the more attractive concepts in regenerative therapy is stem cell transplantation of enriched or purified tissue-derived stem cells, or in vitro manipulated embryonic stem cells. This therapy has the potential to restore the partial loss of organ function. The ultimate goal of regenerative therapy is to develop fully functioning bioengineered organs that can replace lost or damaged organs following disease, injury or aging. We have recently developed a method for creating three-dimensional bioengineered organ germ, which can be used as an ectodermal organ such as the tooth or whisker follicle (Nature Methods 4, 227-30, 2007). Our analyses have provided a novel method for reconstituting this organ germ and raised the possibility of tooth replacement with integrated blood vessels and nerve fibers in an adult oral environment. However, it remains to be determined whether a bioengineered tooth can achieve full functionality.
Outline of the research outcome
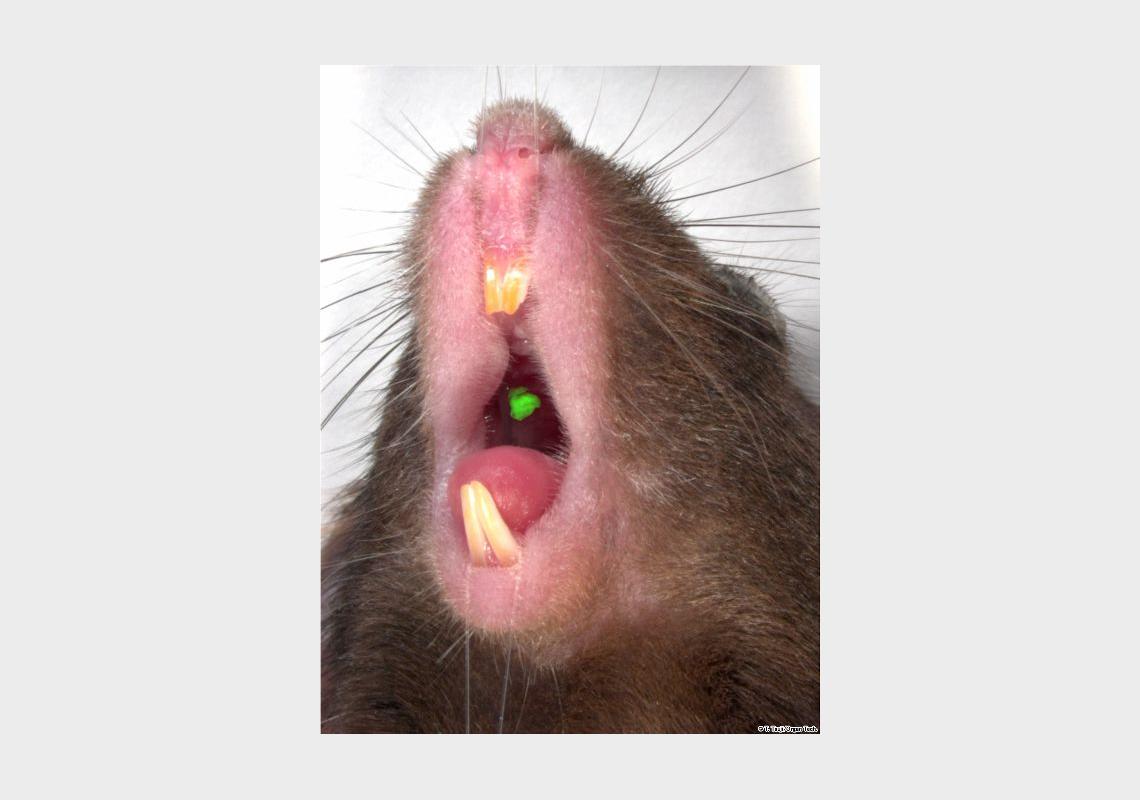
We analyzed the development of a bioengineered tooth germ transplanted into adult oral environment. To develop a tooth regenerative method for a clinical application, a bioengineered tooth should achieve full functionality, including sufficient masticatory performance, biomechanical cooperation with tissues in the oral and maxillofacial regions, and proper responsiveness via sensory receptors to noxious stimulations in the maxillofacial region. Recently, we developed an adult murine lost tooth transplantation model. After the extraction of first molar, its cavity was repaired by osteogenesis for a month. Then, a hole was prepared in alveolar bone using drill. Then, a bioengineered molar tooth germ was transplanted into the hole with correct orientation and gingival transplantation area was sutured (Fig. 1a, b).
1. The eruption and masticatory potential of the bioengineered tooth
At 16 days after transplantation, eruption of the bioengineered tooth could not be observed. At 37 days after transplantation, exposure of the cusp tip occurred in the gingival area of transplantation, indicating an eruption of the bioengineered tooth. At 49 days and thereafter, this bioengineered tooth was observed to have erupted to reach the occlusal plane and was seen to achieve opposing tooth occlusion.
Following the achievement of occlusion, there was no excessive increase in the tooth length at up to 120 days after transplantation (Figure. 2). However, the crown width of the bioengineered tooth was smaller than that of other teeth, since at present we are not able to regulate the crown width and cusp position. We are now trying to develop a novel cell manipulation technology to regulate the tooth size and morphology.
We next performed a Knoop hardness test, which is a test for mechanical hardness. The hardness of these mineralized tissues of the bioengineered tooth, not only enamel but also dentin, were equivalent with those of normal tooth. The microhardness test suggested that the bioengineered tooth have sufficient masticatory performance as well as the normal, mature tooth
2. Bioengineered tooth response to mechanical stress
It is well known that an alveolar bone remodeling was induced via the response of periodontal ligament according to a mechanical stress such as the treatment of orthodontic movement. Previous studies demonstrated that the localizations of osteoclast for bone absorption in compression side and osteoblast for bone formation in tension side were observed. Thus, we analyzed the tooth movement and localizations of osteoclast and osteoblast for bone remodeling in alveolar bone by an experimental orthodontic movement. A
normal tooth and the bioengineered tooth was moved buccally for 17 days with a mechanical force. At 6 days after the treatment, histological analysis revealed that morphological changes of periodontal ligament were observed in both the lingual tensioned side and buccal compressed side. The bioengineered tooth could successfully moved in response to the mechanical stress as well as normal tooth
3. Perceptive potential of neurons entering the tissue of the bioengineered tooth
Perception of noxious stimulations such as mechanical stress and pain, are important for the protection and proper functions of teeth. Neurons in the trigeminal ganglion, which innervate the pulp and PDL, can detect these stress events and transduce the corresponding perceptions to the central nervous system. In our current experiments, we evaluated the responsiveness of nerve fibers in the pulp and PDL of the bioengineered tooth to induced noxious stimulations. Nerve fibers were detected in the pulp, dentinal tubules, and PDL of the bioengineered tooth as in a normal tooth. We also found in our current analyses that the nerve fibers innervating both the pulp and PDL of the bioengineered tooth have perceptive potential for nociceptive stimulations and can transduce these events to the central nervous system (the medullary dorsal horn).
In this study, we successfully demonstrated that our bioengineered tooth germ develops into a fully functioning tooth with sufficient hardness for mastication and a functional responsiveness to mechanical stress in the maxillofacial region. We also show that the neural fibers that have re-entered the pulp and PDL tissues of the bioengineered tooth have proper perceptive potential in response to noxious stimulations such as orthodontic treatment and pulp stimulation. These findings indicate that bioengineered tooth generation techniques can contribute to the rebuilding of a fully functional tooth. Our study provides the first evidence of a successful replacement of an entire and fully functioning organ in an adult body through the transplantation of bioengineered organ germ, reconstituted by single cell manipulation in vitro. Our study therefore makes a substantial contribution to the development of bioengineering technology for future organ replacement therapy
Research Institute for Science and Technology, Tokyo University of Science
●Adress: 2641 Yamazaki, Noda, Chiba, 278-8510, JAPAN
●Project Leader: Takashi Tsuji (Professor of the Research Institute for Science and Technology, Tokyo University of Science and Director of Organ Technologies Inc.)
●Homepage: http://www.tsuji-lab.com/
■ Organ Technologies Inc.
●Adress: 2-2 Kandatsukasamachi, Chiyoda, Tokyo, 101-0048, JAPAN
●President: Hiroaki Asai
●Homepage: http://www.organ-technol.co.jp/
■ Health Labor Sciences Research Grant from the Ministry of Health, Labor and Welfare
“Research on Regenerative Medicine for Clinical Application”
・Research Domain Leader: Akira Yamaguchi (Professor, Oral Pathology, Department of Oral
Restitution, Division of Oral Health Sciences, Graduate School, Tokyo Medical and Dental University)
■ Grants-in-Aid for Scientific Research Project of the Ministry of Education, Culture, Sports,
Science and Technology “Scientific Research for Priority Area: System cell engineering (bio-engineering) by multiscale manipulation”
・Research Domain Leader: Toshio Fukuda (Professor, Institute for Advanced Research / Graduate School of Engineering, Nagoya University)
■ Grants-in-Aid for Scientific Research Project of the Ministry of Education, Culture, Sports,
Science and Technology “Scientific Research (A)”
・Principal investigator: Takashi Tsuji (Professor of the Research Institute for Science and Technology, Tokyo University of Science and Director of Organ Technologies Inc.)