Changes in Characteristics of Membrane Currents.
Professor Byung-Chang Suh’s research team from the Department of Brain and Cognitive Sciences at Daegu Gyeongbuk Institute of Science and Technology (DGIST), South Korea, succeeded in identifying a new operational mechanism principle of the ‘Acid-sensing Ion Channel,’ which recognizes internal pain in an organism.
The findings are expected to have a significant impact on further studies focusing on the development of therapeutic agents that control pain by providing a more precise understanding of the operational mechanism of the ‘Acid-sensing Ion Channel’ that plays a pivotal role in transmitting pain signals.
Pain is transmitted to the brain through nociceptive nerves when pain spots that are distributed within an organism are stimulated. Precisely, when pain causing substances are coupled to the plasma membrane of the cells that constitute the pain spots, the pain signals are recognized.
Inside the organism, changes in pH levels occur under pathophysiological conditions such as inflammation, ischemia, cancer, and the like, which are accompanied by pain. The Acid-sensing Ion Channel (ASIC) detects changes in pH levels in the organism and transmits the pain signal to the brain. Biologically, many studies have been conducted regarding the Acid-sensing Ion Channel; however, many areas are still unclear, especially in terms of the operational mechanism and the cell membrane merging mechanism.
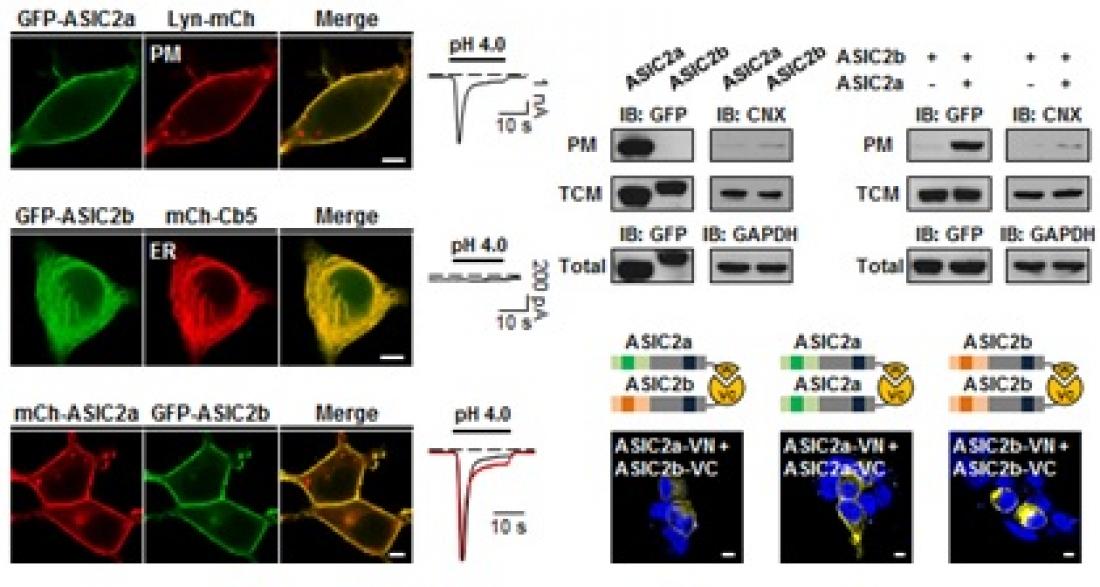
Professor Suh’s research team detected the cell membrane merging mechanisms that modulate the activity of the Acid-sensing Ion Channel at the molecular level, and it is this discovery and identification of the new cell membrane merging mechanism of the Acid-sensing Ion Channel that had remained unknown until now.
The research team identified through animal experiments that there is a different cell membrane merging mechanism between subunits of the Acid-sensing Ion Channel. ASIC2a can be merged to cell membranes and has a cell membrane merging signal in protein, unlike ASIC2b, which cannot be merged to cell membranes. In addition, ASIC2b has no cell membrane merging signal and can only be merged to the cell membrane by forming a heteromeric complex with ASIC2a.
The outcome of this study is meaningful since it identified a new cell membrane merging mechanism of the Acid-sensing Ion Channel, and furthermore is significant that it proposed a research direction for a new understanding of the activity control mechanism of various ion channels among subunits including ASIC2, ASIC2b and many other subunits.
Professor Byung-Chang Suh from DGIST’s Department of Brain and Cognitive Sciences said, “Understanding of the cell membrane merging and activity control mechanism of the Acid-sensing Ion Channel plays an important role in identifying the pain signal transmission system. That our study investigated a new control mechanism of the Acid-sensing Ion Channel has important implications. Through continuous research, I’ll strive to identify additional operations that act on the nervous system and will develop new ways to treat pain.”
This research outcome was published in the online edition of Scientific Reports, a sister publication to the international academic journal Nature, on August 1, 2016. The study was conducted with the support of junior executive researchers at the Ministry of Science, ICT, and Future Planning; the Center for Nerve Aging and Regeneration Research at DGIST; and the Center for Cerebral Cortex Research at the Korea Brain Research Institute.