Organic Letters
About Organic Letters
Organic Letters rapidly publishes short reports on creative, innovative organic synthesis, from catalysis to natural products to functional materials.
- Website: https://pubs.acs.org/journal/orlef7?ref=breadcrumb
- Current Issue: https://pubs.acs.org/toc/orlef7/current
News
31 Jan 2022
Ehime University
Aromaticity of the Space Surrounded by Two Aromatic Rings
23 Jan 2022
Ehime University
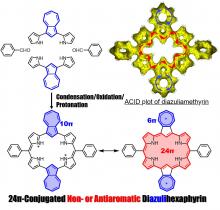
A ring-expanded porphyrin, diazuliamethyrin, was successfully synthesized via a “3+3” condensation method. This porphyrin showed a 24pi non- or antiaromatic character. We analyzed the optical and electronic structures using magnetic circular dichroism spectroscopy and time-dependent density functional theory calculations.
25 Jun 2021
Ehime University
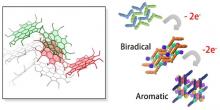
A ring-expanded porphyrin with no meso-bridges comprised of an odd number of pyrroles was successfully synthesized via the oxidative coupling of the corresponding terpyrrole. This porphyrin showed a 34pi aromatic character and an intense absorption at the near-infrared region. We analyzed the optical and electronic structures using magnetic circular dichroism spectroscopy and time-dependent density functional theory calculations.
17 Oct 2019
Ehime University
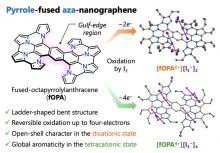
Polycyclic heteroaromatic molecules are a highly useful class of organic materials. In this study, synthesis of a new azacoronene, in which both pyrrole and azulene moieties are circularly fused, was achieved in just three steps. This new azacoronene exhibited multistep reversible oxidations under electrochemical and chemical conditions. Formation of an aromatic 22π-electron conjugation and a tropylium cation (6π-electron conjugation) in the dicationic state played a key role in stabilizing this cationic species.


![Selective Synthesis of Cyclo[9]pyrroles Based on an Oxidative Coupling](https://www.asiaresearchnews.com/sites/default/files/styles/medium/public/articles_images/image1_7.jpg?itok=JpWY_x68)