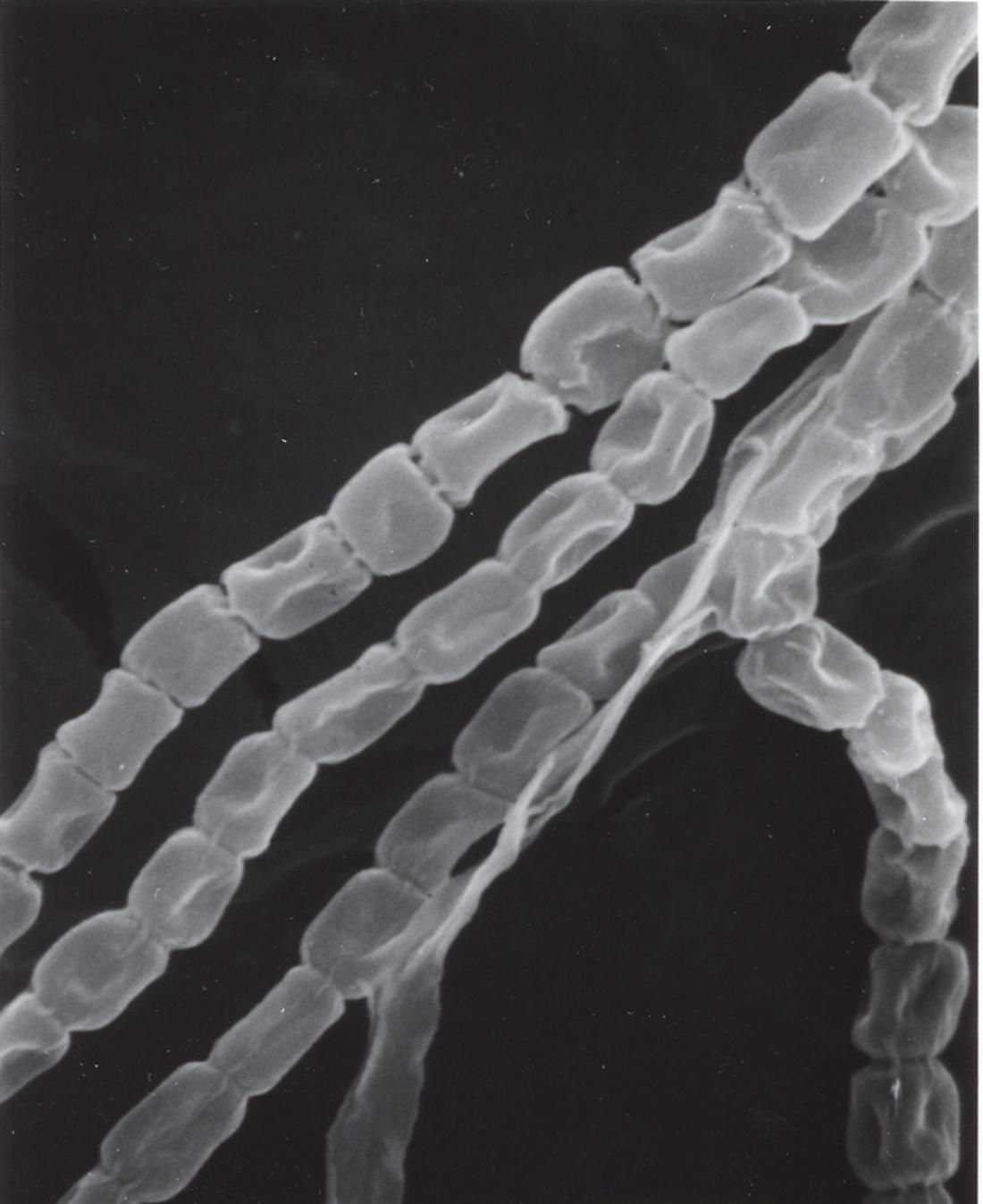
An electron micrograph of the bacterium Streptomyces sp. SN-593.
Some of the most promising drugs in the clinical pipeline are derived from compounds that microbes already produce naturally. With a time-scale of millions of years, their evolution-powered drug development process may not be the most efficient in terms of speed, but the resulting molecules can achieve remarkably specific and potent interactions with important biological targets.
The bacterium Streptomyces sp. SN-593, for example, produces the antibiotic reveromycin A (RM-A), explains Shunji Takahashi, a senior research scientist with Hiroyuki Osada’s team at the RIKEN Advanced Science Institute in Wako. “RM-A was originally isolated as an inhibitor of cancer cell proliferation,” says Osada. RM-A is also a potent inhibitor of metastatic tumor growth in bone—it induces cell death specifically in cells called osteoclasts, which makes it a promising anticancer drug candidate.
RM-A is one of a larger family of compounds known as spiroacetal polyketides, which are typically manufactured via a complex, multi-enzyme process. Synthesis of these compounds proceeds in the fashion of an assembly line, with each enzyme introducing a particular chemical modification to a precursor molecule before passing it along to the next enzyme.
By understanding the nuts and bolts of this process, scientists can potentially ‘hack’ production to manufacture more effective drugs. Osada, Takahashi and colleagues recently made important strides in this regard by identifying the enzymes that generate RM-A’s distinctive core ‘spiroacetal’ structure.
Touring the factory floor
Previous attempts to dissect RM-A biosynthesis based on the analysis of individual candidate genes have run into signficant obstacles. “In most cases, we determine the physiological substrates of one enzyme in an unknown biosynthetic pathway by disrupting its gene and analyzing the accumulated biosynthetic intermediate,” says Takahashi. Unfortunately, Streptomyces sp. SN-593 can be a difficult organism to manipulate genetically, and the production intermediates are too chemically unstable to isolate and characterize.
Fortunately, the researchers were able to devise a set of techniques that enabled them to comprehensively analyze the genetics and biochemistry of RM-A production in this organism. They began by cultivating Streptomyces under conditions that maximized the activity of the RM-A production machinery. This enabled them to zoom in on a cluster of genes that all participate in this drug assembly line, which they subsequently sequenced. The ‘backbone’ of RM-A is assembled over the course of a 62-step process by multiple gene products containing specialized functional domains that collectively form a complex known as the polyketide synthase (PKS) system, after which another set of enzymes introduces various post-PKS modifications that ultimately yield the final drug product.
Based on their analysis of the various enzymes in the PKS pathway as well as a series of experiments with isotope-labeled RM-A precursors, Osada, Takahashi and colleagues were able to identify a key intermediate in the synthetic pathway, which they termed RM-A1a. Oddly, this intermediate contains a propionyl group, a small three-carbon ‘tail’ that is added during synthesis but is notably absent from the final target molecule. “We have not yet solved the physiological significance of this additional propionyl unit which must later be truncated,” says Takahashi.
RM-A1a is a linear molecule that must subsequently be rendered circular, a process known as ‘cyclization’ that the researchers hypothesized would be enacted by an enzyme known as a dehydrogenase. They identified such an enzyme in their gene cluster, and demonstrated that disruption of the revG gene, which encodes this protein, leads to the accumulation of RM-A1a. Experiments combining RM-A1a with purified RevG confirmed that this enzyme facilitates the transition of this RM-A precursor into a cyclization-ready intermediate.
Getting oriented
Just like humans, molecules can be right- or left-handed. This property of ‘handedness’, also known as chirality or stereochemistry, depends on the relative positioning of their various chemical groups, and can radically affect a molecule’s biological properties. Osada, Takahashi and colleagues were surprised to note that the intermediate generated by RevG ultimately gives rise to a mix of products of varying stereochemistry, even though the naturally occurring RM-A product only exhibits ‘15S’ stereochemistry. “We had thought that the formation of the final spiroacetal stereochemistry would be non-enzymatically selected, but rather based on [differences in] thermodynamic stability,” says Takahashi.
This hinted at the action of a yet-unidentified enzyme, and gene-disruption experiments revealed the protein RevJ as the missing link. Without RevJ, Streptomyces cells produced 75% less ‘15S’ RM-A than their wild-type counterparts. Accordingly, the researchers were able to show that the sequential action of both RevG and RevJ is required to drive formation of the appropriate spiroacetal structure during RM-A synthesis.
According to the researchers, by unearthing the secrets of production in this way, it should become possible to develop drug derivatives in a more rational fashion, rather than simply stumbling through a process of trial and error. “Most chemical modifications of RM-A result in the loss of its bioactivity,” says Takahashi. Finding ways to bolster the stability of RM-A without undermining its overall capacity for preventing bone destruction will be a primary objective. He and Osada intend to ‘fill in the gaps’ in the production process as a means to expand their chemical modification palette.
In parallel, the researchers also hope to determine the coordinating mechanisms that enable all of these various biosynthetic enzymes to work together in harmony. “We will solve the regulatory mode of gene expression in the RM-A cluster,” says Takahashi.
Reference:
Takahashi, S., Toyoda, A., Sekiyama, Y., Takagi, H., Nogawa, T., Uramoto, M., Suzuki, R., Koshino, H., Kumano, T., Panthee, S. et al. Reveromycin A biosynthesis uses RevG and RevJ for stereospecific spiroacetal formation. Nature Chemical Biology 7, 461–468 (2011).