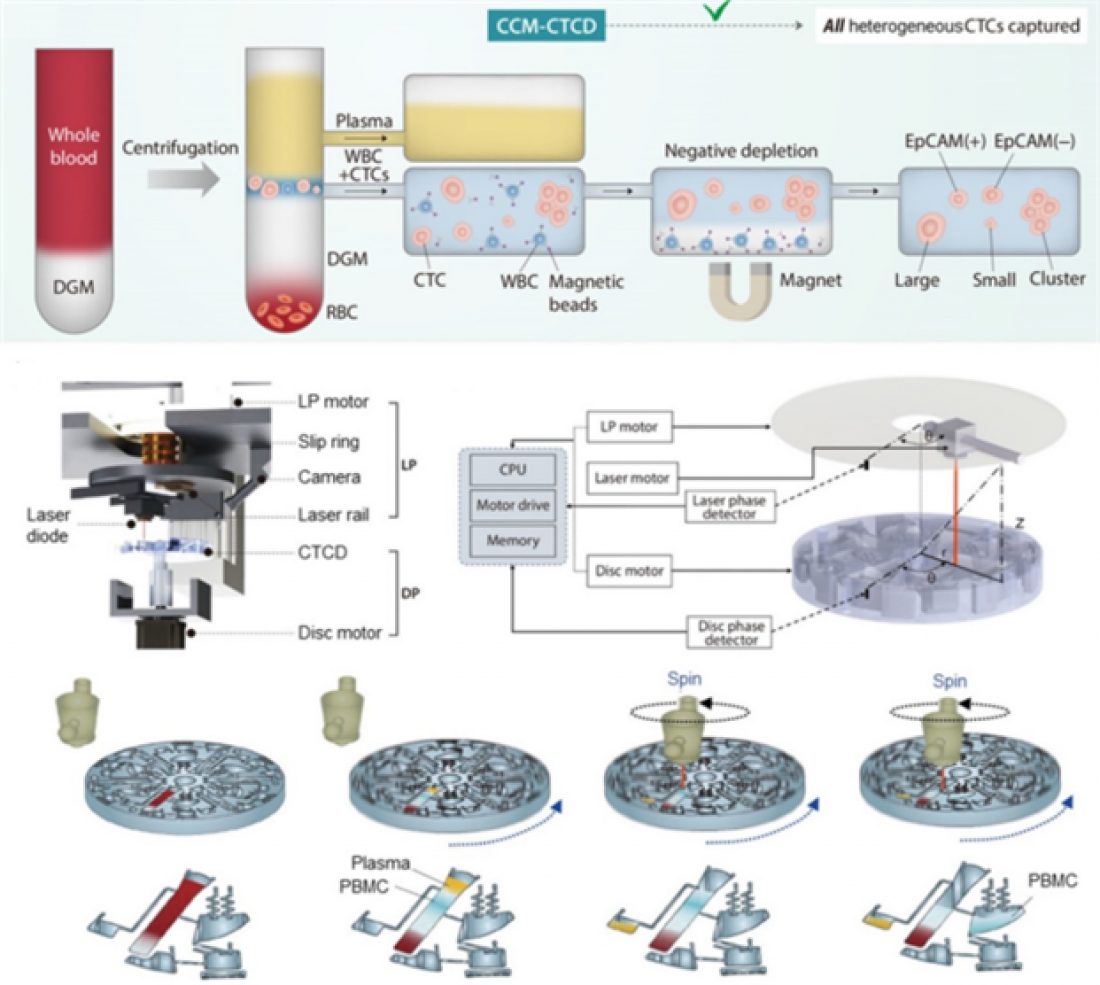
A new approach developed by DGIST scientists and CTCELLS company teams in Korea can successfully and rapidly isolate rare circulating tumor cells from patient blood samples. The team’s findings, published in the journal Theranostics, could help improve cancer diagnosis and the ability to provide targeted and personalized treatments.
Circulating tumor cells are cells that break off from cancers and are released into the blood stream. They can go on to form the seeds for new tumor formation in other parts of the body, known as metastases. The advantage of isolating them from the blood is that they represent the diversity of cancer cells found in a person’s body and identifying them could lead to more targeted therapies. But current techniques used to do this either miss some types of circulating tumor cells or are done manually, which takes a lot of time and specialized training.
“Our smart and practical approach realizes a big dream in the field of liquid biopsy and demonstrates high performance across a wide range of cell types and different cancers with full automation,” says Minseok S. Kim at the Department of New Biology at Daegu Gyeongbuk Institute of Science & Technology (DGIST).
The fully automated centrifugation approach is called Continuous Centrifugal Microfluidics – Circulating Tumor Cell Disc (CCM-CTCD). It involves placing a tube containing a blood sample in a machine with a spinning disc. The spinning, or centrifugal force, causes the blood to separate into layers containing different components, with the heavier red blood cells dropping to the bottom, lighter cells floating in a middle layer, and plasma settling at the top. After the disc begins to spin, a laser motor starts rotating at the same angular velocity and phase. This crucial step allows a laser to move and open a valve in the blood sample tube while the disc continues to spin, maintaining a thin layer of tumor and white blood cells, which are released into a separate chamber. The chamber contains antibodies that specifically attach to and separate white blood cells from the mixture. This allows the circulating tumor cells in the mixture to flow on their own into a final chamber.
The team was able to go on to identify the different types of tumor cells and confirm by subsequent DNA testing that they represent the full diversity of different types of cells in the blood sample. They also used the technique on blood samples from patients with varying stages of lung cancer and found the number of circulating tumor cells in a sample correlated with the stage of disease progression. Identification of the types of tumor cells also allowed them to modify treatment strategies.
The team is now working on commercializing the technique for clinical use and hope to expand its application to isolate other types of cells, including nerve, stem and immune cells.
Corresponding author’s email: [email protected]