Klebsiella pneumoniae is a major bacterial pathogen that can cause serious infections, especially in patients with compromised immune system. And it can easily spread through healthcare facilities, such as neonatal intensive-care units. It belongs to the Klebsiella genus which is classified by the World Health Organization as one of the three “critical priority pathogens” for its multi-drug resistance and hence new drugs are urgently needed. *
In 2017, Professor Chen Sheng, who then worked at the Hong Kong Polytechnic University, together with his research team and collaborators from Zhejiang University, discovered a new strain of Klebsiella pneumoniae which was not only resistant to carbapenem – a highly effective antibiotic commonly used for treating severe or high-risk bacterial infections, but also hyper-virulent and highly transmissible. Their findings were published in Lancet Infectious Diseases (DOI: 10.1016/s1473-3099(17)30489-9).
A puzzle to be solved
“But we did not know how this superbug was formed,” said Professor Chen, who is now the Acting Head of Department of Infectious Diseases and Public Health at City University of Hong Kong.
To solve the puzzle, Professor Chen and his team continued the investigation of this new hyper-virulent Klebsiella pneumoniae strain.
Recently, when they isolated a carbapenem-resistant Klebsiella pneumonia strain from a patient in a Chinese hospital, the phylogenetic analysis revealed that the strain actually belonged to Klebsiella variicola strain. “Klebsiella variicola was rarely isolated from clinical samples. It usually resides in the respiratory and gastrointestinal tract of patients without producing typical signs of bacterial infections,” said Professor Chen.
The team discovered that this Klebsiella variicola strain actually carries a new plasmid - a ring-shaped piece of DNA, which harbours the key genes of the virulence plasmid commonly found in hyper-virulent Klebsiella pneumoniae, leading to high-level virulence and making the bacteria more dangerous.
In the experiments, when the mice were infected with carbapenem-resistant Klebsiella pneumonia with this plasmid, all the mice died, and the mortality rate was similar to the hyper-virulent control strain. But when the mice were infected with the same strain which lacked this plasmid, all of the mice survived.
The secret behind the superbug
Further experiments by the team also discovered that the new-found plasmid can travel from one bacterium to another. The transfer of this virulence plasmid to a carbapenem-resistant Klebsiella pneumoniae strain can result in a significant increase in the virulence level of such strain, making it both carbapenem-resistant and hyper-virulent.
The new findings were published in Nature Microbiology end of last month (DOI: 10.1038/s41564-019-0566-7).
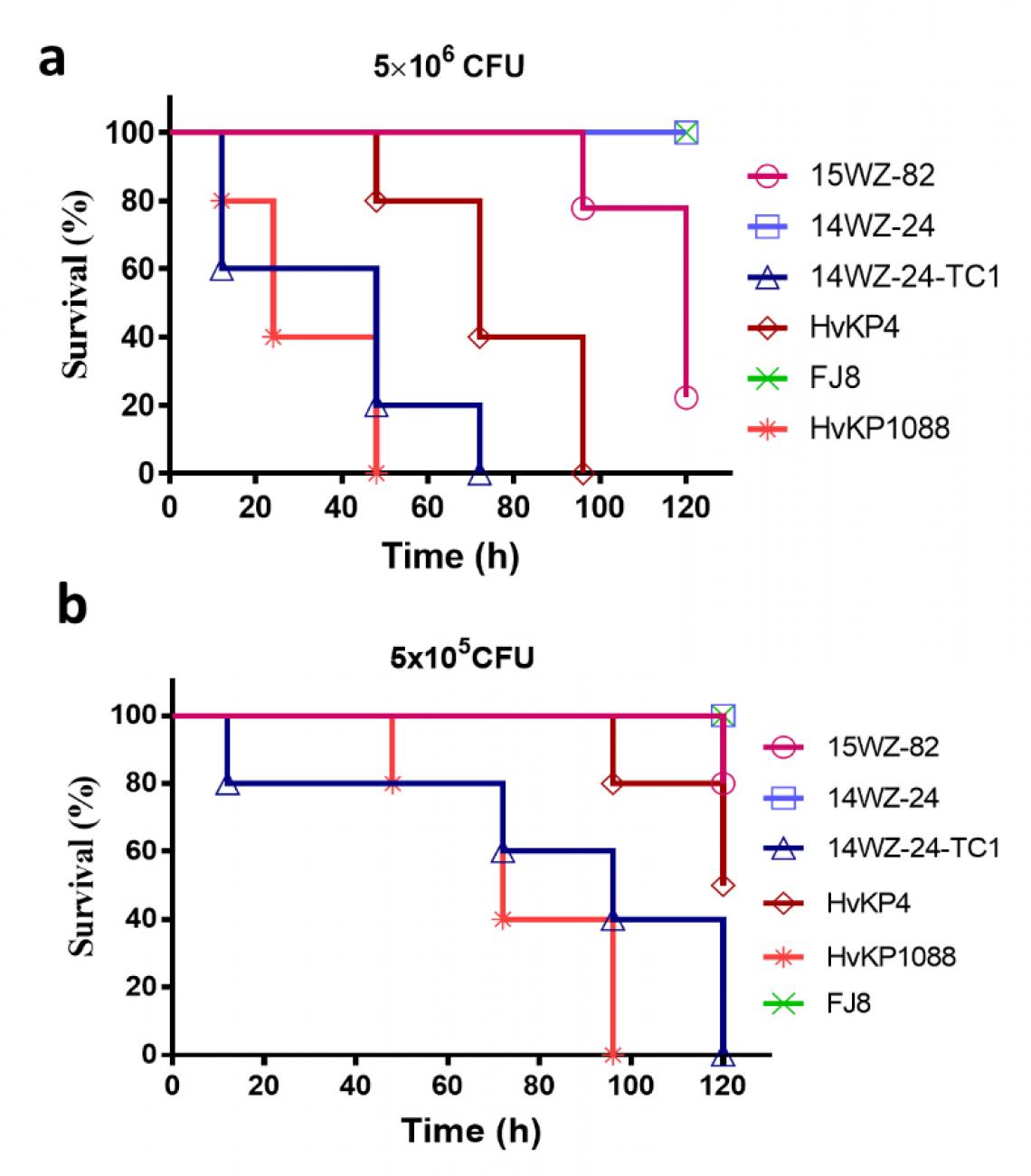
Professor Chen and his team carried out experiments to compare the survival rates of the mice infected with different bacterial strains and different dosages. Testing strains included the Klebsiella variicola strain 15WZ-82 isolated from a patient, classic ST11 Klebsiella pneumonia strain 14WZ-24, ST11 carbapenem-resistant and hypervirulent Klebsiella pneumoniae strain 14WZ-24-TC1 (i.e. the one carrying plasmid from 15WZ-82), HvKP4 and HvKP1088 (both hypervirulence control), as well as classic carbapenem-resistant Klebsiella pneumoniae strain FJ8 (low-virulence control).
Fig a (infection by a higher bacterial dose) shows that although 15WZ-82 exhibited a lower virulence level than the control HvKP strain (mortality rate of the mice infected with 15WZ-82 strain after 120 hours is 80%, while the mice infected with HvKP4 all died after 96 hours), such virulence level was nevertheless much higher than the classic ST11 carbapenem-resistant Klebsiella pneumoniae strain.
Fig b (infection by a lower bacterial dose) shows that the mice infected with 14WZ-24-TC1, i.e. classic ST11 carbapenem-resistant Klebsiella pneumoniae strain carrying plasmid, all died after 120 hours, a level slightly lower than that of hyper-virulent control strain HvKP1088 but far higher than HvKP4; on the contrary, the mice infected with the same dose of 14WZ-24 (classic ST11 Klebsiella pneumonia strain 14WZ-24 which does not carry the plasmid) all survived.
Credit: Nature Microbiology (DOI: 10.1038/s41564-019-0566-7)
Professor Chen Sheng worries that the newly discovered conjugative plasmid could covert a normal Klebsiella pneumoniae strain into a hyper-virulent Klebsiella pneumoniae in a rapid way, posing a bigger
threat to patients' health.
Photo credit: City University of Hong Kong
“The significance of the current study is that we discovered a new conjugative plasmid, which may be responsible for the formation of the new type of superbug that we discovered in 2017. This conjugative plasmid could covert a normal Klebsiella pneumoniae strains into a hyper-virulent Klebsiella pneumoniae in a very quick way. This suggests that the hyper-virulent Klebsiella pneumoniae, which can cause a deadly infection in humans, could become prevalent due to the transmission of this plasmid in clinical Klebsiella pneumonia and pose a bigger threat to patients’ health,” said Professor Chen.
Based on this finding and more data from his team, Professor Cheng said that there would be more different types of conjugative virulence plasmid transmitting around in clinical Klebsiella pneumoniae. His team will continue to investigate more clinical Klebsiella penumoniae strains to discover more different types of conjugative virulence plasmids, which attribute to the current emergence of hypervirulent and carbapenem-resistant Klebsiella pneumoniae.