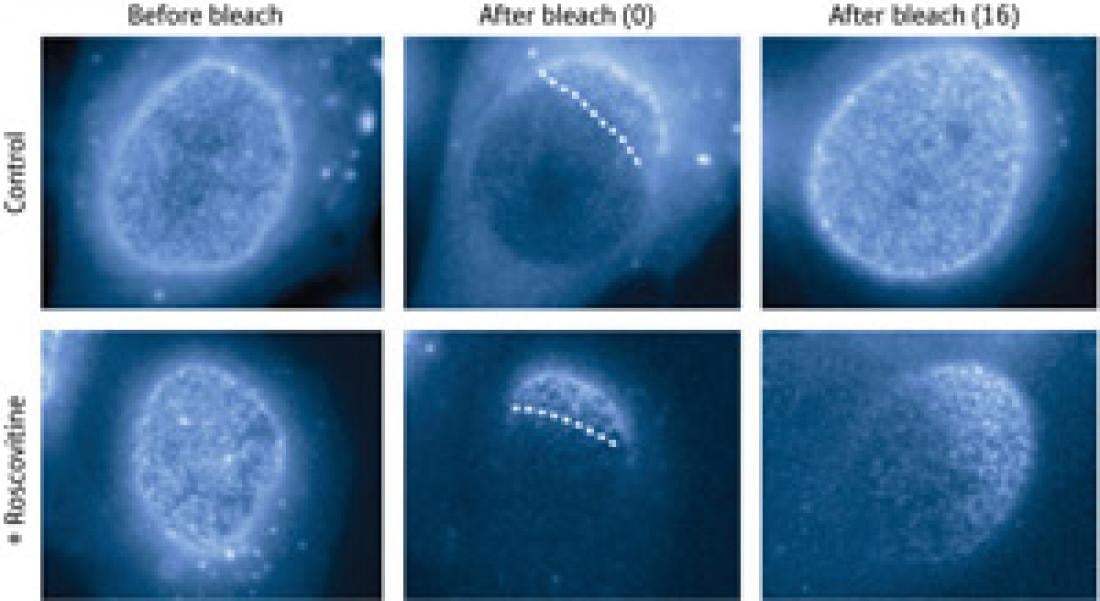
Figure 1: Interphase NPC formation can be visualized using a laser photobleaching method that involves fluorescently tagged Nups (central panels). The fluorescence of the bleached area recovered after 16 hours incubation in control cells (top right), whereas addition of the CDK inhibitor roscovitine prevented the recovery (bottom right).
A team led by Naoko Imamoto of the RIKEN Advanced Science Institute in Wako has uncovered processes governing the formation of functionally important structures called nuclear pore complexes (NPCs) in dividing human cells1.
Mitosis, the process of mammalian cell division, is followed by a period called ‘interphase’ during which the volume of the cell nucleus almost doubles; as does the number of NPCs on the nuclear envelope separating the ‘nucleoplasm’ from the cytoplasm—the rest of the cell’s contents.
Innumerable molecules shuttle between the nucleus and cytoplasm through pores formed by NPCs, which are large octagonal structures composed of multiple copies of around 30 different proteins called ‘nucleoporins’ (Nups). NPCs form at the end of mitosis, when previously disassembled NPCs are reassembled, and during interphase when their number increases in preparation for another round of cell division.
“We wished to understand how NPCs form on the nuclear envelope of interphase cells, which is much less understood than post-mitotic NPC formation initiating on mitotic chromosomes,” says Imamoto.
The researchers began by developing fluorescence-based NPC visualization methods. Their first approach involved using a laser to photobleach certain nuclear surface areas of early human interphase cells expressing fluorescently tagged ‘scaffold’ Nups. This allowed them to monitor the formation of new NPCs, which appeared as bright dots in the bleached areas.
A second method involved monitoring NPC formation in fused cells called ‘heterokaryons’. “Our heterokaryon method allows quantitative analysis of many nuclei without risking laser damage,” explains Imamoto.
By combining these visualization techniques with cell engineering experiments they found that the formation of NPCs in human interphase cells is promoted by known cell-cycle regulators called cyclin-dependent protein kinases (Cdks).
Cdk inhibition experiments further revealed that CdK1 and CdK2, in particular, govern NPC formation during interphase, but not in post-mitotic NPC assembly, suggesting that different regulatory mechanisms are at play. Interestingly, Cdks also govern the behavior of nuclear envelope proteins, suggesting an as yet unknown mechanistic connection between NPC formation and membrane dynamics.
Cdks appear to act early in NPC formation because their inhibition suppressed the generation of small ‘nascent’ pores observed on early interphase nuclear envelopes using scanning electron microscopy.
“We believe that these ‘nascent pores’ are probably immature nuclear pores, although this needs to be confirmed,” says Imamoto.
Cdk inhibition did not greatly affect nuclear growth, suggesting that its mechanism of regulation is distinct from that of NPC formation, which is something that future research must also address.
The corresponding author for this highlight is based at the Cellular Dynamics Laboratory, RIKEN Advanced Science Institute