Gears are an essential component of everyday machines. The ability to shift gears, like in a car, allows for control of the degree or direction of motion generated, making machines more versatile. Now, a team led by researchers at the Institute for Chemical Reaction Design and Discovery (WPI-ICReDD) in Hokkaido University has reported a new design strategy for realizing molecular-sized gears in crystals and the first example of controllable molecular gear shifting in a solid material. They developed a crystalline material that contains gear-like molecules which can be reversibly shifted between two types of motion. The design principle provides a blueprint for the development of versatile, new materials.
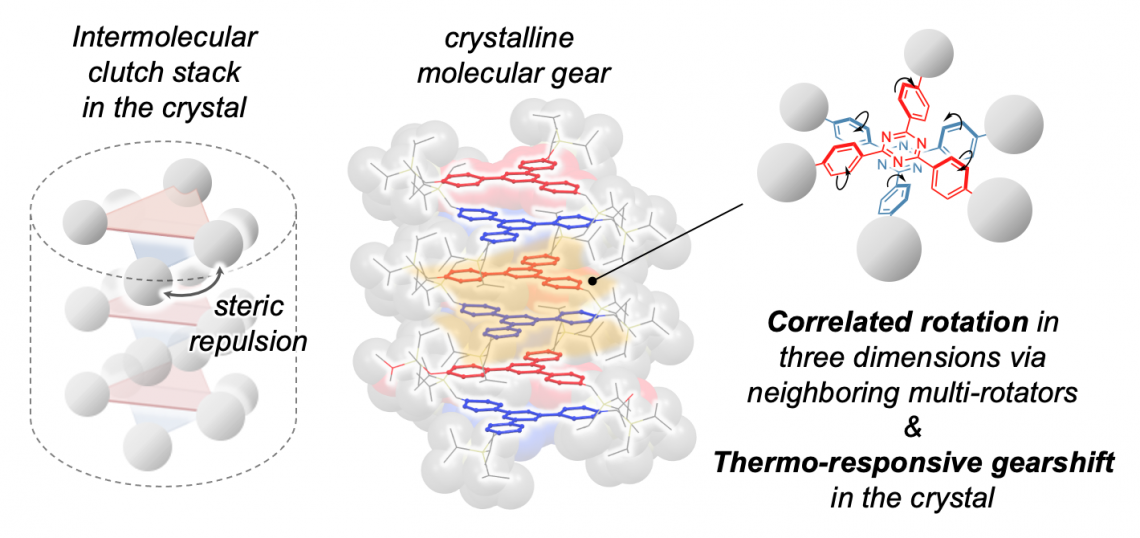
Researchers utilized a gear-shaped molecule called triaryltriazine, which has a center triazine ring with three phenylene rings attached to it—which act like the teeth of a gear. By attaching bulky, stationary molecules to the phenylene rings, researchers induced a “clutch stack” arrangement, where adjacent triaryltriazine molecules are rotated 60° from each other, rather than stacking in the same orientation.
“The design of the clutch stack was inspired by the mechanical machinery system of the clutch in a car,” said Associate Professor Mingoo Jin.
(Left) Schematic of the clutch stack arrangement. (Middle) Structure of the molecular gears in the clutch stack. (Right) Rotational direction of two adjacent molecular gears. (Mingoo Jin, et al. Journal of the American Chemical Society. December 7, 2023)
The attached stationary molecules also created enough space for the three phenylene rings to rotate between two positions in a flapping motion. The clutch stack arrangement of the triaryltriazine molecules enabled adjacent molecules to hook on to each other as the phenylene rings rotated, much like interlocking gears. This resulted in the correlated motion of all the molecules in the stack.
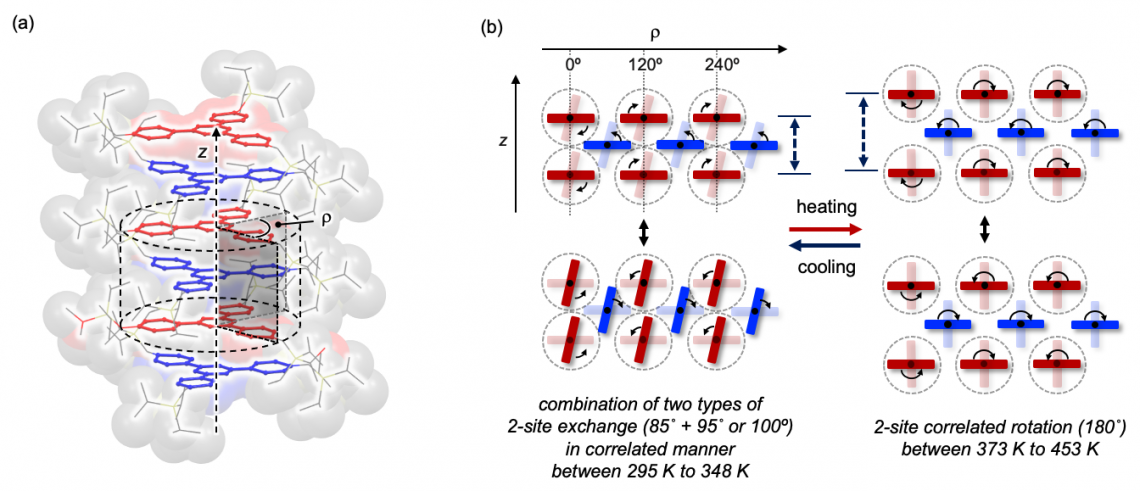
When the temperature was raised above a certain threshold, a different correlated motion was observed, in which phenylene rings underwent a 180° rotation. This change in motion was attributed to a phase transition in the crystal that created more space between adjacent molecules, giving the phenylene rings more room to rotate.
(a) Structure of the molecular gears in the clutch stack. (b) Side view of molecular gears showing the two modes of motion observed at lower temperatures (left) and higher temperatures (right). (Mingoo Jin, et al. Journal of the American Chemical Society. December 7, 2023)
Researchers found this change in motion could be reversed by cooling the crystal, marking the first time such controllable molecular motion has been observed in a solid. The effect of the molecular gearshift could be fine-tuned by adjusting the size and structure of the stationary molecule attached to the gear molecule. This adjustability opens the door to the development of new functional materials that leverage crystalline molecular machines.
“The next direction for our research would be using geared molecular motion in crystals to manipulate different physical properties of solid-state materials, such as light emission or thermal behavior” commented Jin.
Members of the research team at the Institute for Chemical Reaction Design and Discovery (WPI-ICReDD), Hokkaido University. Left to right: Tetsuya Taketsugu, Mikhail Tsitsvero, Mingoo Jin, Andrey Lyalin, Hajime Ito. (Photo: WPI-ICReDD)