DGIST Professor Jaeheung Cho in the Department of Emerging Materials Science at DGIST (up), Bohee Kim, a Combined M.S-Ph.D. Program Student in the Department of Emerging Materials Science (below left), and Donghyun Jeong, a Combined M.S-Ph.D. Program Student.
DGIST research team succeeded in synthesizing new biomimetic materials that will increase the efficiency of chemical reaction related to body metabolism and discovered that synthesized materials cause the oxidation of aldehydes. The results are expected to bring positive impacts on catalyst development in the future.
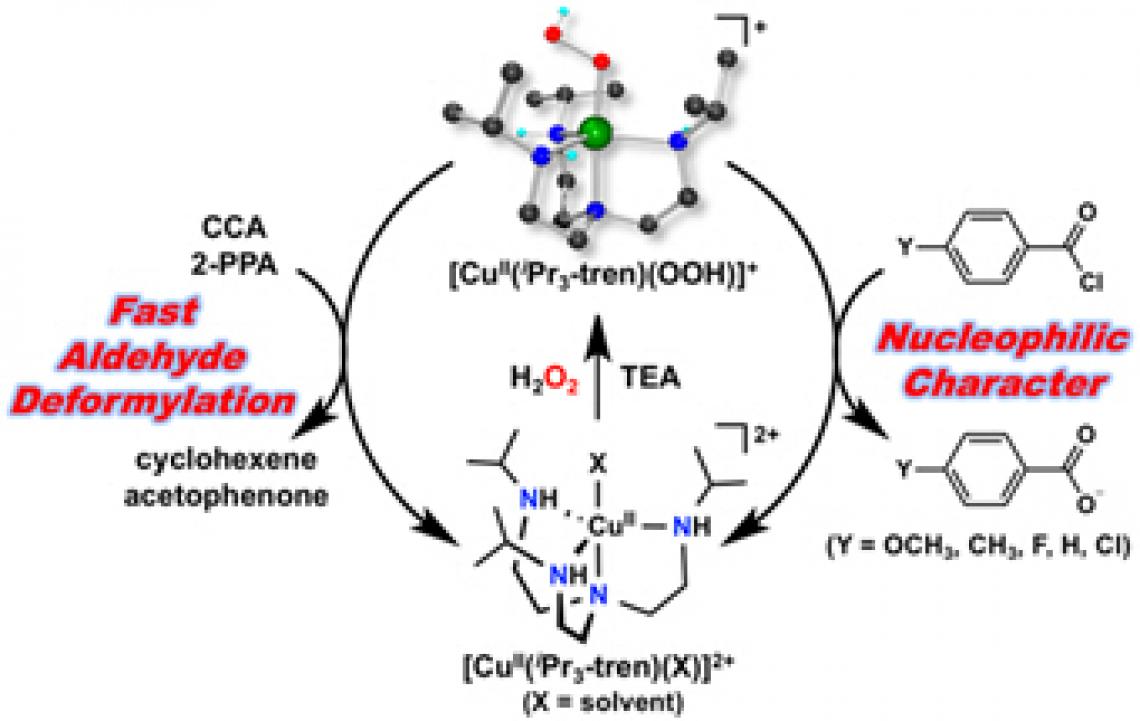
DGIST announced that Professor Jaeheung Cho’s team in the Department of Emerging Material Science developed ‘copper(II)-hydroperoxo1 complex,’ a biomimetic compound that conducts aldehyde deformylation. This complex is drawing attention in bioinorganic fields due to its higher efficiency than the existing complexes which cause deformylation reaction.
‘Deformylation reaction’ is a type of oxidation2 reaction that deformylates double-bond structure of carbon and oxygen inside aldehyde or ketone3 compound in a different form. It especially adjusts important chemical reaction in various ways by controlling the conversion of steroid with sex hormone or by converting the fat aldehyde formed during fat reduction4. To synthesize biomimetic compounds, the research team added ‘hydroperoxo,’ a combination of electrons and proton with oxygen molecule, to a copper compound. The ‘copper(II)-hydroperoxo complex’ formed here boasts much higher efficiency in deformylation reaction than other existing materials.
It is especially the first time to confirm that copper(II)-hydroperoxo adduct is directly involved in deformylation reaction.
Professor Jaeheung Cho in the Department of Emerging Materials Science at DGIST said “We believe we’ve expanded the research scope of materials involved in deformylation reaction, which plays an important role in biological system. We will continue our research on materials or method that have higher efficiency and controllability in deformylation reaction,” and explained his follow-up research plans.
This research was conducted with support by the Ministry of Science and ICT, National Research Foundation of Korea, the C1 Gas Refinery R&D Center, Basic Research Project (mid-level researchers), and the Magnetics Initiative Life Care Research Center. The result was published July 18 on Communications Chemistry, a sister journal of the world-class international journal Nature.
1 Hydroperoxo: One photon is combined with a ‘peroxo’ which has two electrons added to an oxygen molecule
2 Oxidation: Chemical combination with oxygen or hydrogen
3 Ketone: Hydrocarbon derivatives connected with two carbonyl functional groups
4 Reduction: Elimination of oxygen from an oxide