Mechanisms of molecular pathology and erythromycin treatment in myotonic dystrophy type 1
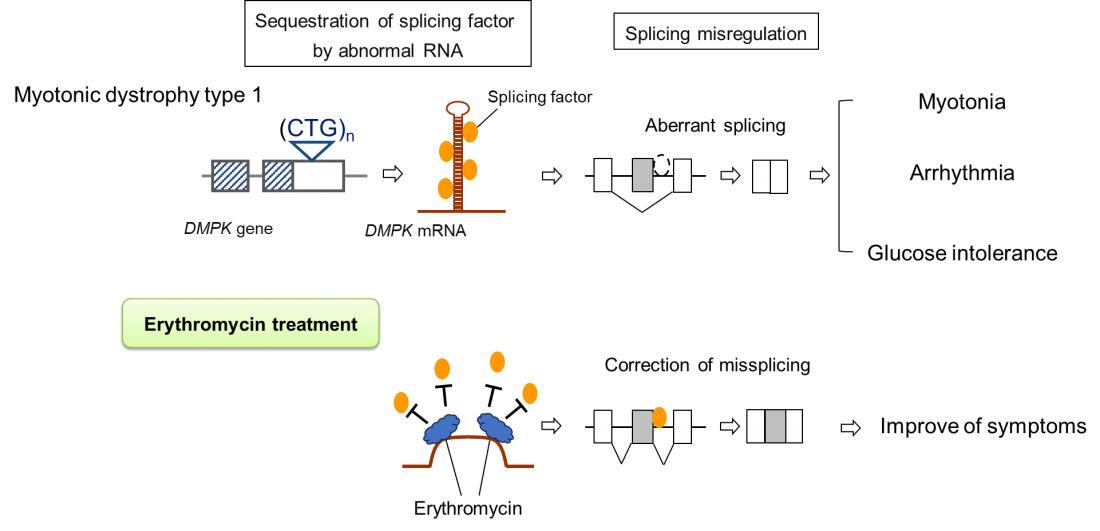
In myotonic dystrophy type 1, aberrant RNA transcribed from the mutant DMPK gene leads to the aggregation of splicing regulators. The resulting depletion in intracellular splicing regulators disrupts splicing control mechanisms, and splicing abnormalities occur in various genes. These splicing abnormalities are believed to cause diverse symptoms throughout the body, including myotonia, progressive muscle weakness, arrhythmias, and impaired glucose tolerance.
In myotonic dystrophy type 1, erythromycin prevents the aggregation of splicing regulators by binding to the abnormal RNAs, thereby restoring splicing regulatory function and improving symptoms.
Researchers from Osaka University find that the antibiotic erythromycin has acceptable safety and tolerability profiles for treating myotonic dystrophy type 1, and are optimistic about its efficacy
Osaka, Japan – Myotonic dystrophy type 1 (DM1) is a genetic disease characterized by progressive muscular weakness. There is currently no treatment, despite many recent efforts. But now researchers from Japan may have found a cure.
In a study published on line in eClinicalMedicine, researchers from Osaka University have reported an innovative approach to identify a possible new treatment for DM1, which has just passed phase 2 clinical testing with flying colors.
DM1 is the most common form of muscular dystrophy. It is caused by a genetic mutation that affects an essential process known as alternative splicing, which is important for the correct assembly of proteins all over the body. In this way, DM1-causing mutations affect many different bodily systems, leading to a wide range of symptoms that are difficult to treat.
To identify potential therapies for DM1, the research team used drug repositioning screening, a method in which drugs that are already used for a specific purpose are tested for their effects in other diseases. They found that erythromycin—which you may already be familiar with, given its widespread use as an antibiotic—may be effective in DM1 because of its potential for improving splicing abnormalities.
“After our successful preclinical trials in DM1 cell and mouse models, we had high hopes for erythromycin,” says Masayuki Nakamori, lead author of the study. “As the next step in getting this drug to the clinic, we conducted a trial on 30 people with DM1 to study the treatment’s safety and tolerability, with a secondary aim of investigating its efficacy.”
The researchers gave 6 of the 30 patients a placebo, while 12 received a low dose (500 mg) and 12 received a high dose (800 mg) of erythromycin for 24 weeks. Because these long-term doses are already used for other diseases, the researchers hoped that the safety data would be positive—and fortunately, they were right.
“We were also very interested in testing the efficacy of erythromycin in patients with DM1,” explains Masayuki Nakamori. “To do this, we mainly looked at markers of aberrant splicing directly in patients’ muscle biopsies.”
Although this efficacy testing revealed few significant results, the research team noted that several patients who received erythromycin showed major improvements in splicing markers, suggesting that some patients respond better than others. Further analysis also revealed that some splicing markers were significantly improved at one dose of erythromycin but not the other, indicating that dosage may be important.
Despite the small sample size, the researchers are very optimistic. Erythromycin continues to be a promising therapeutic option, and subsequent phase 2b and 3 trials should be performed as soon as feasible to see whether this widely used antibiotic can successfully treat DM1, or at least a subset of patients.
About Osaka University
Osaka University was founded in 1931 as one of the seven imperial universities of Japan and is now one of Japan's leading comprehensive universities with a broad disciplinary spectrum. This strength is coupled with a singular drive for innovation that extends throughout the scientific process, from fundamental research to the creation of applied technology with positive economic impacts. Its commitment to innovation has been recognized in Japan and around the world, being named Japan's most innovative university in 2015 (Reuters 2015 Top 100) and one of the most innovative institutions in the world in 2017 (Innovative Universities and the Nature Index Innovation 2017). Now, Osaka University is leveraging its role as a Designated National University Corporation selected by the Ministry of Education, Culture, Sports, Science and Technology to contribute to innovation for human welfare, sustainable development of society, and social transformation.
Website: https://resou.osaka-u.ac.jp/en