C1 polymerization of diazoacetates with new Pd-based initiating systems
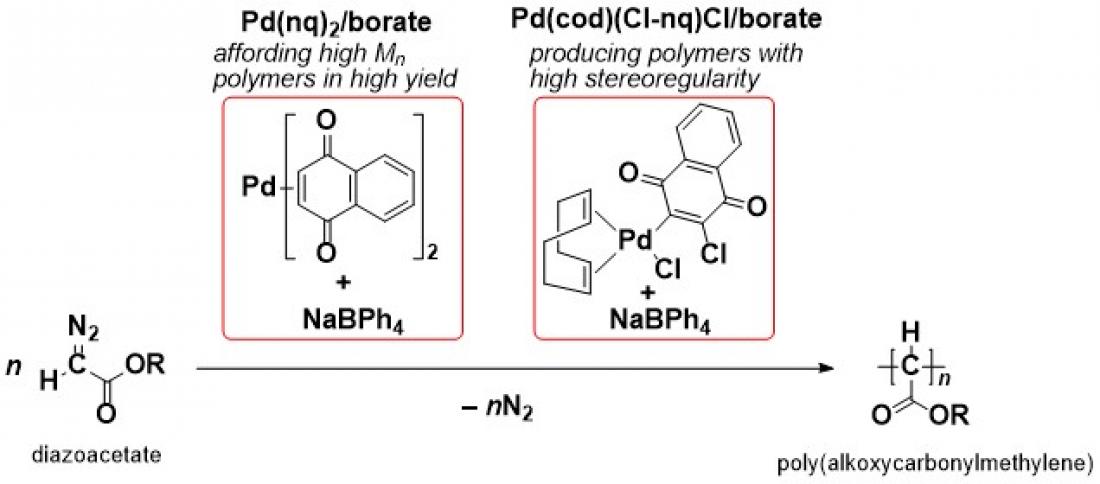
A research team in Ehime University found that new initiating systems consisting of palladium, naphthoquinone, and borate exhibited unique activities for C1 polymerization of diazoacetates, affording high molecular weight poly(alkoxycarbonylmethylene)s in high yields or regulating the stereostructure of the resulting polymers. A detailed investigation of the initiating systems provided some important information about the Pd-initiated polymerization of diazoacetates, which would be beneficial for the development of Pd-based initiating systems with further improved activities.
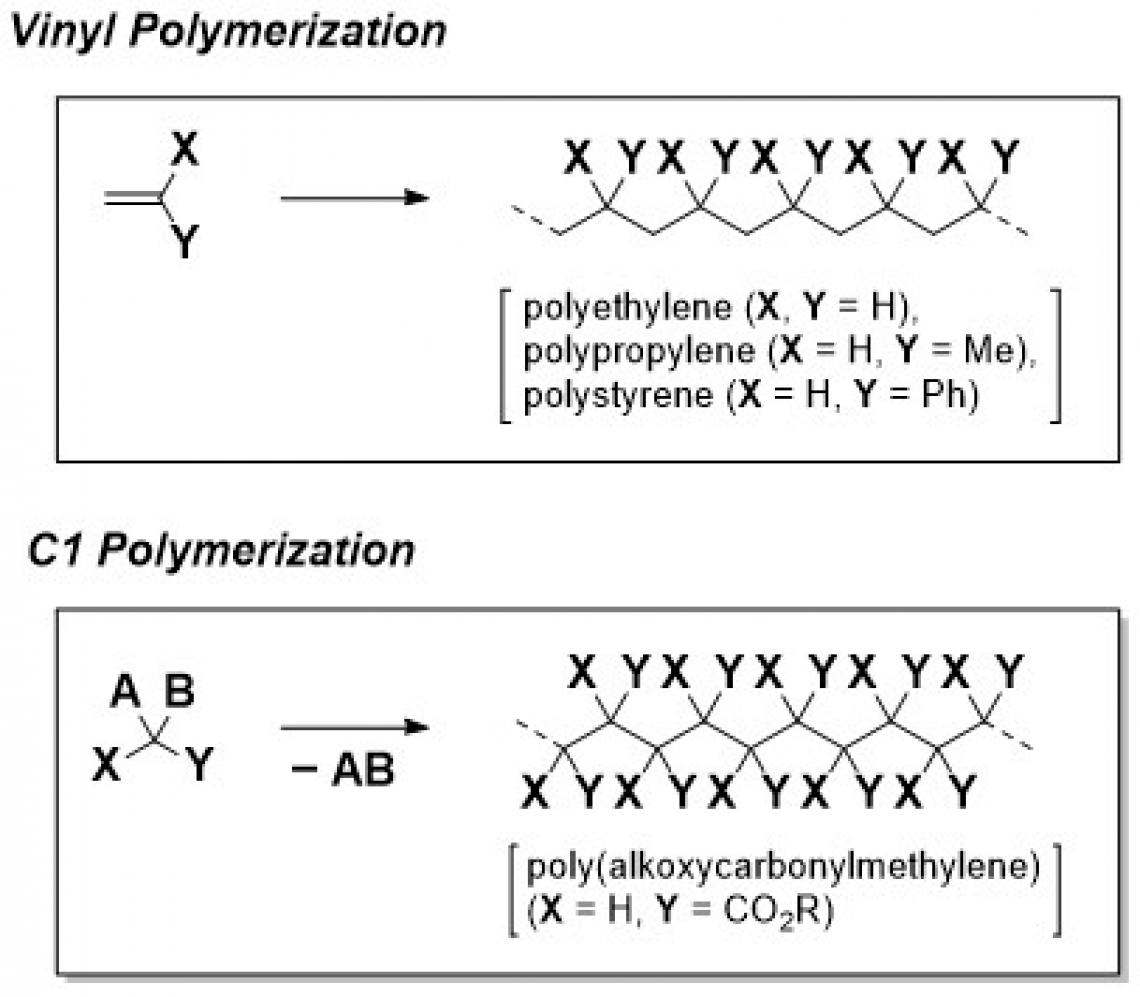
C1 polymerization is a method for preparing polymers with a carbon-carbon (C-C) main chain from “one carbon unit”, in contrast to conventional vinyl polymerization, which constructs a C-C main chain from a “two carbon unit” and has been utilized for the production of a variety of polymers with industrial importance such as polyethylene, polypropylene, and polystyrene. and so forth. The most important characteristic of C1 polymerization is that it can yield polymers, bearing a substituent on each main chain carbon atom, which are expected to possess unique physical properties and/or functionalities but are generally difficult to prepare by vinyl polymerization. Accordingly, C1 polymerization has attracted much attention in the last few decades, and the research team, directed by E. Ihara and H. Shimomoto, has been leading the research field since 2003 with their first paper on the polymerization of diazoacetates initiated with Pd-based systems in 2003, where the group demonstrated for the first time (independent of a Chinese group almost at the same time) that diazoacetates were suitable monomers for C1 polymerization, giving C-C main chain polymers with an alkoxycarbonyl group on each main chain carbon atom [poly(alkoxycarbonylmethylene)s]. Even though the research group has had important success in originally developing Pd-based initiating systems such as (NHC)Pd/borate [NHC = N-heterocyclic carbene, borate = NaBPh4] and ?-allylPdCl/borate for the polymerization of diazoacetates, there still remains much to be improved, with respect to molecular weights, yields, and stereoregularity of the resulting polymers.
In this paper, the research group reported two newly prepared initiating systems consisting of palladium, naphthoquinone (nq), and borate; one is Pd(nq)2/borate, and the other is Pd(cod)(Cl-nq)Cl/borate [cod = 1,5-cyclooctadiene, Cl-nq = 2,3-dichloro-1,4-naphthoquinone]. The former system, Pd(nq)2/borate, exhibited high activity for the polymerization of diazoacetates, affording polymers with higher molecular weights in higher yields than those of the products obtained with the previously reported Pd-based initiating systems. On the other hand, the latter initiating system, Pd(cod)(Cl-nq)Cl/borate, was capable of yielding polymers with a highly stereoregulated structure, even though the polymer yield was relatively low. In addition to these unique activities of the new initiating systems, the research group succeeded in revealing some important mechanistic aspects, including polymer chain end structures with high resolution mass spectrometry and a molecular structure of the precursor complex, Pd(cod)(Cl-nq)Cl, with an X-ray crystallographic analysis.
The results reported in this paper are of significant importance for further improving the activity of Pd-based initiating systems for the C1 polymerization of diazoacetates.