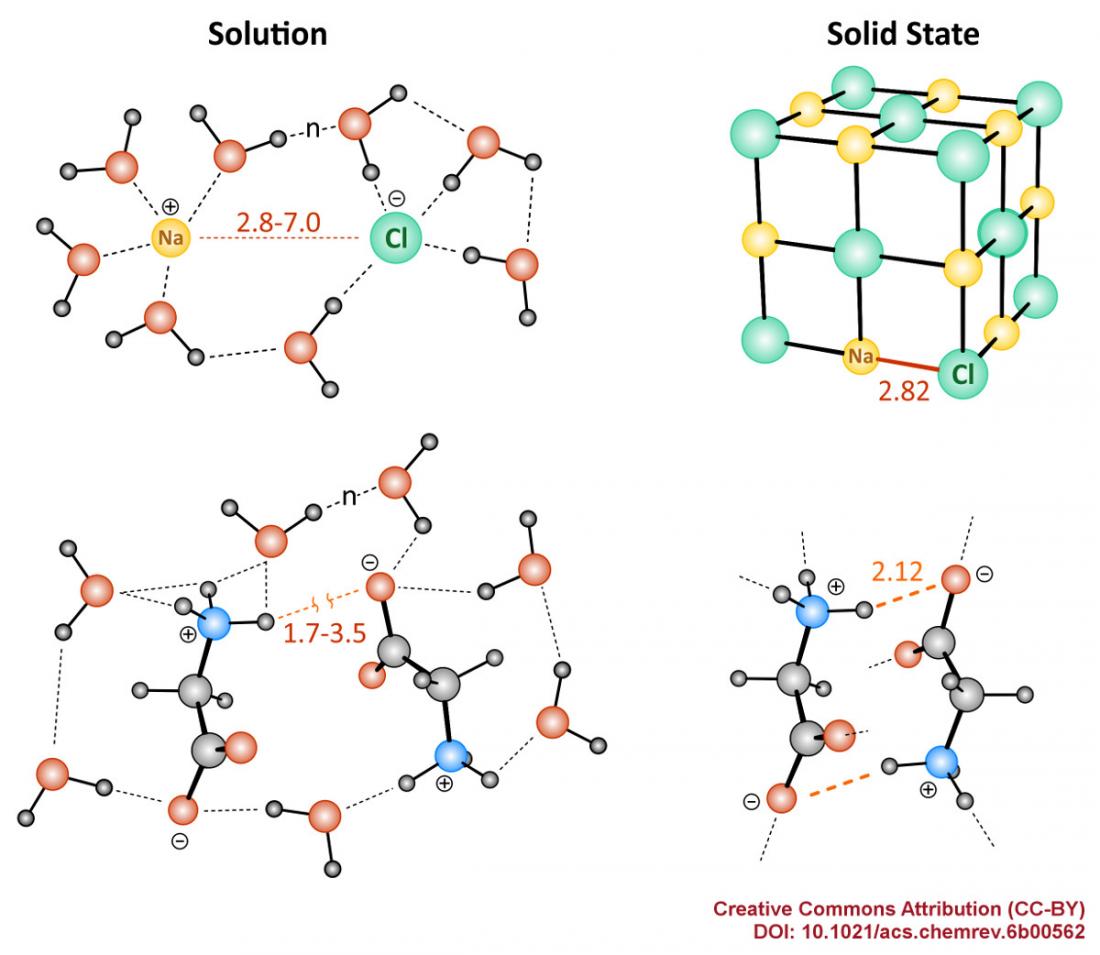
Figure: Different behavior of inorganic and organic ionic molecules in solid state and in solution
Covalent and ionic bonds are the two major types of chemical bonding in molecular structures and complex molecular architectures. The covalent bond involves sharing of electron pairs between atoms, while the ionic bonding is based on the electrostatic attraction between oppositely charged ions. A critical difference between the covalent and ionic bonding is exhibited upon changing from solid to liquid phase systems. Covalent compounds usually change their structure and electron properties only slightly upon dissolution in water and other liquids. In contrast, in ionic compounds solvent-solute interactions induce dramatic changes upon dissolution, and the properties of the resulting liquid phase system depend strongly on the media (see Figure).
In a recent analysis of state-of-the-art research efforts in the area of ionic compounds, a new emerging trend has been identified as IOLIOMICS, a group led by Valentine P. Ananikov at Russian Academy of Sciences and report in their review paper. The acronym is a combination of [IO]ns, [LI]quids and [OMICS], where the suffix -omics refers to describing the comprehensiveness of the available data. IOLIOMICS is defined as a research discipline dealing with the studies of ions in liquids (or liquid phases) and stipulated with fundamental differences of ionic interactions.
Studies on ionic interactions drive the development of many demanding applications in biology, chemistry and material science. Very promising directions with a proven practical potential include, among many others, ionic channels and ion control in living cells, ionic drugs and drug delivery systems, ion batteries, ionic liquids, electrolytes. Complex ionic systems are challenging for structural and mechanistic studies in solution, and none of the currently available analytic methods can offer a complete characterization. Dynamic behavior and flexible nature of ion-solvent assemblies induce another dimension of structural variations. Thus, a demanding requirement of comprehensive data analysis is of urgent need.