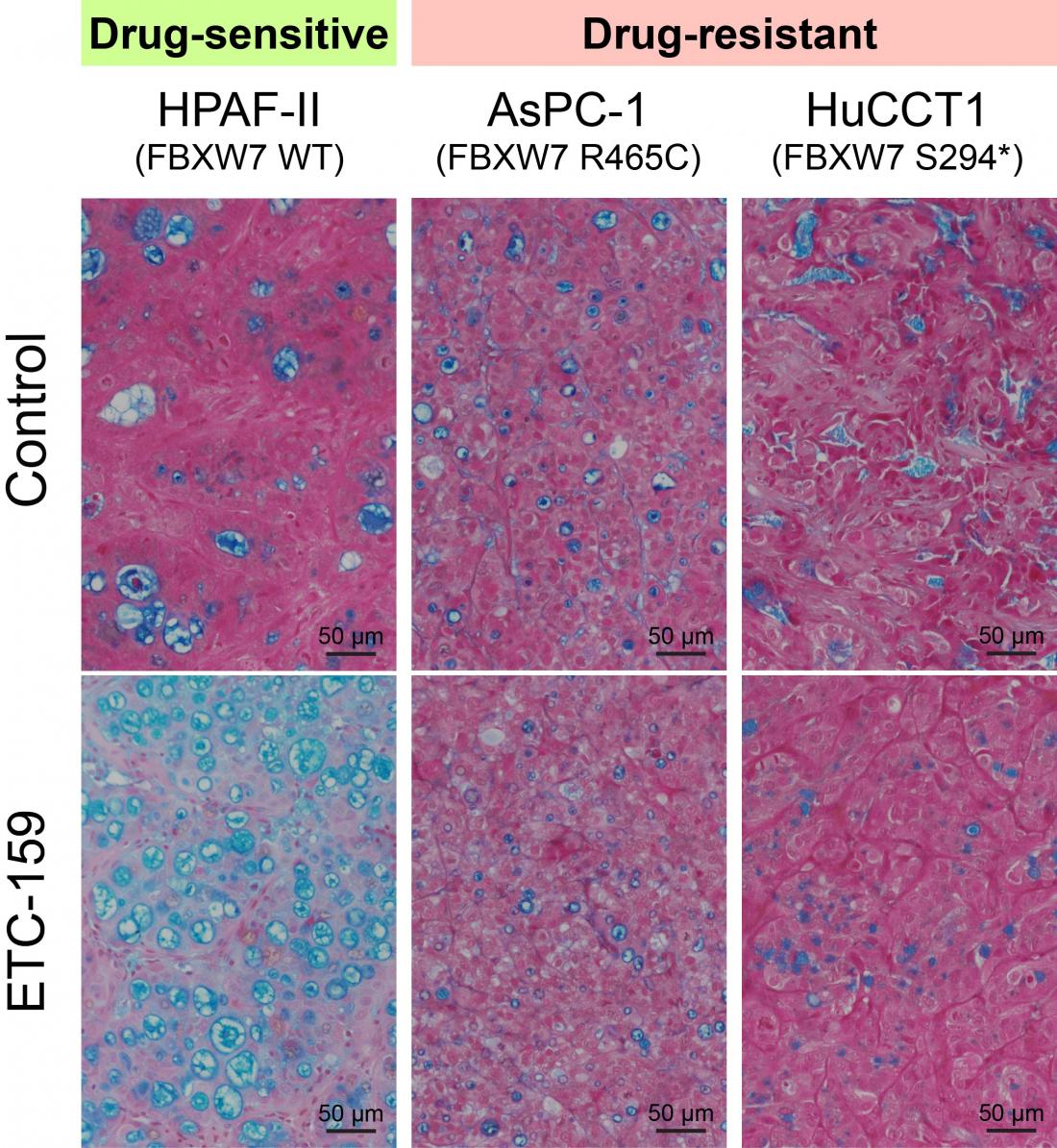
Duke-NUS scientists show that FBXW7-mutant cancers are resistant to Wnt inhibitors by comparing the impact Wnt inhibitor ETC-159 has on cancer cells with and without FBXW7 mutation. In cancer cells with non-mutated FBXW7, administering ETC-159 forces fast growing cancer cells to develop into more mature cells (measured by the level of mucin expression, which is captured in blue), slowing their growth and the spread of the tumour (left column). However, when the gene is mutated (middle and right columns), the inhibitor has no such effect.
Scientists at Duke-NUS Medical School have uncovered why some pancreatic and colorectal cancers[1] fail to respond to Wnt inhibitors, a promising new class of cancer drugs currently under development for these cancers. Their discovery, published in Science Advances, not only offers a new cancer therapy target but also a potential screening tool to identify those patients who will not benefit from this new therapy once it becomes available.
Many gastrointestinal cancers grow uncontrollably when a mutation sets a key biological pathway that governs cell growth, called Wnt, on hyperdrive. The Wnt pathway is hijacked in this way in more than 80 per cent of colorectal cancers and some pancreatic cancers, driving rampant tumour growth. For this group of patients, drugs that block the Wnt pathway, known as Wnt inhibitors, hold great promise and they have been at the centre of intense scientific study, including at Duke-NUS[2].
“Although Wnt inhibitors have shown some promise in certain patients, our study reveals intrinsic resistance in others,” said Dr Zhong Zheng, who led the study as a postdoctoral fellow at Duke-NUS’ Cancer & Stem Cell Biology Programme. “Understanding the mechanisms behind this resistance is crucial for personalised treatments for patients when the drugs don’t slow tumour growth at all.”
Focusing on colorectal and pancreatic cancers with a hyperactive Wnt pathway, Dr Zhong, together with Professor David Virshup who leads the Programme at Duke-NUS, used their Wnt inhibiting drug ETC-159, whose efficacy had been established in preclinical models, to assess the cancer cells’ responsiveness.
By analysing genetics data on both responsive and non-responsive tumours, they discovered that a second mutation in another gene, known as FBXW7, makes cancer cells stubbornly resistant to Wnt-blocking drugs.
FBXW7 mutations occur in about 15 per cent of colorectal cancers. “The FBXW7 mutations change the personality of the cancer,” explained Dr Zhong. “They no longer ‘care’ about the Wnt pathway and so the drugs no longer can do their work.”
Testing tumours for the FBXW7 genetic mutations could spare many patients from receiving ineffective treatment, making this not only a potential biomarker but also as a target for a new type of cancer treatment.
“Predicting drug resistance is critical for precision oncology,” said senior author Prof Virshup. “This work reveals how cancers can evade dependencies on Wnt signalling and serves as a solid foundation for further development.”
“We can now try to target those backup pathways activated by the FBXW7 mutation to overcome the drug resistance,” added Dr Zhong, pointing to new treatment possibilities.
The findings add to previous work by the scientists on how pancreatic cancers become resistant to treatment. Together, these discoveries increase our understanding of the ways cancers find alternate routes to grow and survive.
With more precise therapeutic targets to lock onto, these findings bring the promise of personalised therapies one step closer to reality. In addition to the discovery of FBXW7, the team found these Wnt inhibitor-resistant tumours to be susceptible to an experimental drug known as dinaciclib. Their next step is to investigate the potential of dinaciclib alone and in combination with other agents in treating these cancers.
“Our ultimate goal is to help patients with fully resistant tumours by targeting the alternate cancer pathways unleashed by FBXW7 mutations,” said Prof Virshup. “We hope to translate our findings into more tailored and potent treatment strategies.”
“This research exemplifies the highly translational nature of the basic scientific research conducted at Duke-NUS. Cancers are notoriously diverse, and it is important that we can understand and map that diversity, so that we can offer truly personalised treatment that is effective for the individual and not leave patients to undergo unnecessary therapies that will not work for them,” said Professor Patrick Tan, Senior Vice-Dean for Research at Duke-NUS. “This study is another important step on our journey to make every cancer a treatable disease and the team’s exemplifies our resolve to deliver more effective therapies to patients.”
###
Reference: Zhong Z and Virshup DM. Recurrent mutations in tumor suppressor FBXW7 bypass Wnt/β-catenin addiction in cancer. Science Advances. DOI: 10.1126/sciadv.adk1031
[1] Colorectal cancers are the second most common type of cancer diagnosed in Singapore for both men and women. Pancreatic cancer is the 10th most common cause of cancer in men in Singapore. The World Health Organisation estimated that in 2020 alone, more than 1.9 million new cases of colorectal cancers were diagnosed worldwide.
[2] Duke-NUS, together with A*STAR, developed a made-in-Singapore Wnt inhibitor called ETC-159 which is currently in early phase clinical trials: https://www.duke-nus.edu.sg/about/media/media-releases/media-releases/et...