Figure 1: Elastic electronic circuit made of ionic-liquid-based Bucky gel. In cooperation with Takao Someya and Tsuyoshi Sekitani at the University of Tokyo, Aida and his team are striving to develop a new type of sensor based on elastic electronic circuits made of ‘Bucky gel’. These circuits are worn on the skin and can be used for sweat and temperature measurement. The elastic electronic circuit has great potential for use as skin for robots.
Takuzo Aida
group director
Emergent Materials Department
Responsive Matter Chemistry and Engineering Research Group
RIKEN Advanced Science Institute
“The appeal of chemistry is that you can create new molecules that do not occur in nature,” says Takuzo Aida, Group Director of the Responsive Matter Chemistry and Engineering Research Group launched in October 2007. Aida has embarked on a completely new research project. “An example is the development of special molecular devices similar to living organisms that can respond to different types of stimuli.” What strategies can be used to create molecules with unconventional functions like these? “Like movie actors, molecules are carefully selected to play an important role in creating interesting molecular devices. Thus, a chemist can be compared to a movie director.”
The moment blessed with a discovery
One and a half years into the Aida Nanospace Project (2000–2005), a research program supported by the Exploratory Research for Advanced Technology (ERATO) program of the Japan Science and Technology (JST) Agency, Takanori Fukushima (currently team leader of the Responsive Matter Chemistry and Engineering Research Group’s Functional Soft Matter Engineering Team) placed some powdered carbon nanotubes into an ionic liquid and set the mixture in an ultrasonicator to disperse them. He went out to eat and came back about an hour later. To his surprise, the mixture had turned into a viscous gel similar to mayonnaise. “It has turned into a gel-like substance,” he reported to Aida. Piqued with curiosity, Aida suggested, “Why don’t you poke at it with a pair of tweezers.” The substance immediately returned to its previous state of a powder in suspension. “If Dr Fukushima had returned from eating 30 minutes later, the carbon nanotubes would likely have returned to their original powdered state, and he would have missed the phenomenon. So we believe that we were blessed just one time. Dr Fukushima did not miss the moment,” says Aida.
However, so began a period of difficult work for Fukushima. “I asked him to reproduce the phenomenon, but two months passed without positive results. We thought the gel-forming phenomenon had happened just that once. Dr Fukushima finally said, ‘Let’s stop investigating that phenomenon, it will never happen again.’ However, I persuaded him to continue investigating, since the successful reproduction of the phenomenon would surely lead to the development of a new field of materials. Then three months later, he finally succeeded in finding out how to reproduce the phenomenon.” It was a bold method, involving stirring the solution using a mortar.
Carbon nanotubes are of the order of one nanometer in diameter. They are strong, and also excellent conductors of electricity. Many researchers around the world have attempted in vain to disperse carbon nanotubes uniformly to create new substances. Nanotubes tend to intertwine with each other, preventing them from dispersing uniformly. The problem, however, was solved when the solution was simply mixed and stirred in a mortar. This caused the carbon nanotubes to become disentangled and to disperse completely in the ionic liquid, resulting in the formation of a gel-like substance. Fukushima and his team called the new substance ‘Bucky gel’ (Fig. 1).
A new type of conductive nanotube
Carbon nanotubes are a form of carbon with a graphite structure. Graphite, found commonly in pencil ‘leads’, is composed of stacked carbon sheets. A carbon nanotube is a cylindrical roll of one of these sheets, and carbon nanotubes are the only type of nanotube known to exhibit good electrical conductivity. “We aimed to create a new type of nanotube that could also conduct electricity.”
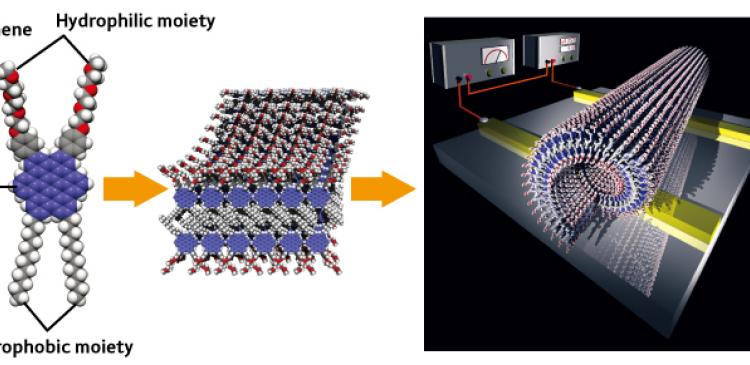
Aida and his team focused on the molecule graphene, which is a small fragment of a single graphite sheet. They created a new type of graphene molecule by adding hydrophilic and hydrophobic moieties, then heated a solution of these molecules to 60 °C. Upon cooling, the molecules combined to form nanotubes of 20 nm in diameter with the hydrophobic moieties on the inside (Fig. 2). “We took advantage of molecular ‘self-organizing’ behavior to create a new type of nanotube.” Self-organization is a mechanism by which molecules combine together naturally to form a complex structure.
Today’s computer circuits are fabricated using light to print very fine structures on hard semiconductor materials such as silicon. This method of forming microscopic structures, based on printing techniques, is generally referred to as a ‘top-down’ method. Improvements in computer performance have relied on continuous enhancement of the top-down method, and efforts have been focused on drawing electrical circuit patterns with ever-finer feature sizes. The top-down method, however, is now reaching its practical limit because the width between wire traces is now less than 100 nm, which is far shorter than the wavelength of the light beam used for processing. Thus, ‘bottom-up’ methods are attracting more and more attention as a means of building up molecules to create fine structures. “The keyword for the bottom-up method is ‘self-organization’. This method allows the fabrication of superfine patterns of several nanometers in width. In other words, it can be used to build up molecules, potential allowing the perfect design of devices.”
The new graphite nanotube is inherently an electrical insulator, but it becomes a conductor when subjected to a simple chemical treatment. Thus, a new electrically conductive nanotube was realized.
“Later we found that the graphite nanotube can still be formed even when the structural design of the original molecule including graphene is modified. In the case of conventional carbon nanotubes, the addition of moieties to the surface causes the tube structure to collapse, and in most cases, also results in degraded electrical conductivity. One of the advantages of graphite nanotubes is the ability to change the structural design of the original molecules, which allows us to specify what new functions are being created.”
For example, Aida and his team, by changing the structure of the original molecule, have successfully created a graphite nanotube that conducts electricity when exposed to light. The expansion of this technique could lead to the construction of highly efficient solar batteries based on organic materials. Conventional solar batteries are mainly made of inorganic silicon material. The use of organic materials will contribute to cost and weight reductions, and will expand the range of application because they are flexible. For these reasons, there is broad interest in the research and development of organic solar batteries. “The energy conversion efficiency and durability of organic solar batteries will not be better than for inorganic solar batteries such as those made of silicon material. Organic materials, however, can be obtained from plants even when oil reserves become depleted. Thus, it is very important for us to establish the basic technology for organic solar batteries because they can be obtained at a low price and resources are inexhaustible.”
Molecular devices activated by two stimuli
Aida launched the Responsive Matter Chemistry and Engineering Research Group at RIKEN in October 2007, and Fukushima assumed the position of team leader of the group’s Functional Soft Matter Engineering Team. “At RIKEN, I do not adopt research themes that I pursued in other research institutes. Instead, I am proceeding with independent research based on new themes. One example is the development of molecular devices that can respond to various types of stimuli, similar to living organisms.”
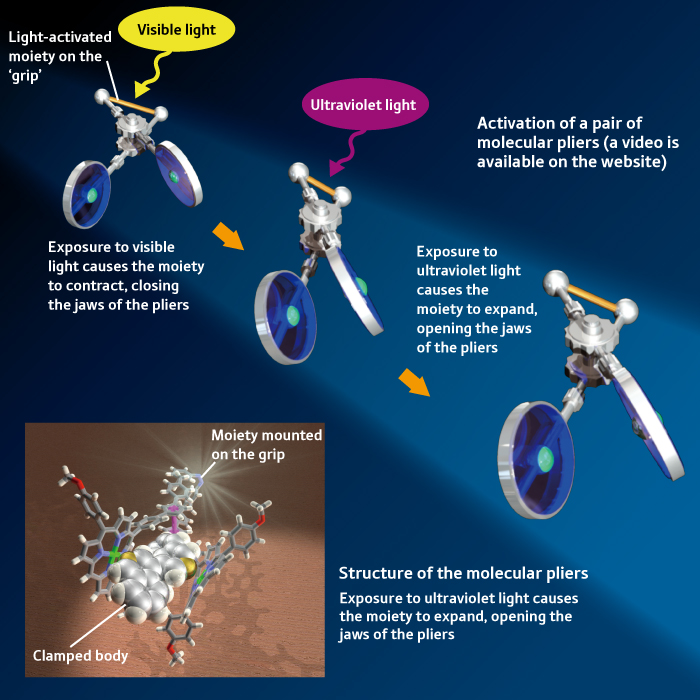
Receptors on the membranes of cells in living organisms differentiate among various signals and open ‘gates’ in response to specific signals in order to take in the necessary materials. “We have also created useful molecular devices that can perform a single function in response to a specific stimulus.” For example, a research laboratory at the University of Tokyo lead by Aida created a pair of ‘molecular pliers’ that are activated by light (Fig. 3). “You cannot hold them of course because the pliers are nano-sized. They have a special moiety, mounted on the ‘grip’, that contracts when exposed to visible light. Using this pair of molecular pliers, we succeeded in clamping and twisting an object.
“We can expand the concept of molecular pliers even further: for example, it could twist when additionally exposed to ultraviolet light, allowing it to twist a clamped object when exposed to ultraviolet light and visible light at the same time, or to twist a clamped object when exposed to ultraviolet light and then to chop if off when exposed to visible light. The device effectively discriminates among various types of stimuli and performs tasks autonomously, similar to a living organism. This is one of the major goals of our project at RIKEN.”
In a laboratory at the University of Tokyo, researchers have already succeeded in creating a new molecular device that can be activated by two stimuli through the modification of a biological molecule called a ‘chaperone’. The functions of proteins are derived from their three-dimensional structure. Thus, they do not function well when their structure collapses. The cylindrical chaperone molecule takes in collapsed proteins and restores them to their original state. It opens its gate and releases the proteins when exposed to an energy carrier called adenosine triphosphate (ATP). “We added to the outside of the chaperone’s gate an artificial gate that opens when exposed to ultraviolet light so that it had a double gate function (Fig. 4). The modified chaperone molecule only opens the double gate completely when exposed to both ultraviolet light and ATP, releasing the restored proteins. By analogy, cars are equipped with a safety system that requires two tasks to be carried out at the same time in order to perform a particular operation. We succeeded in creating a molecular device with this mechanism using the biological chaperone molecule. At RIKEN, we want to use a molecule designed entirely by ourselves for the creation of a device that can be activated by various types of stimuli.”
How can such devices be created? “When you knock over the first in a line of dominoes, it brings down all the other domino pieces in turn after it. This is a good example of an event source affecting the whole system. In an organic material, molecules are arranged in an orderly manner. Thus, we can design the structure of an organic device such that, for example, when a molecule at a certain position is tilted by a certain stimulus, all the other molecules can also be tilted. Furthermore, it is also possible to design the structure of the organic molecular device by controlling the interaction between molecules so that all molecules can be tilted only when the organic material is exposed to two stimuli at the same time. Based on this design concept, we are making efforts to create new molecular devices.”
Focus and patience—creating new molecular devices
“The appeal of chemistry is that you can create new molecular structures that do not exist in nature,” says Aida. “You need to be very focused when creating new molecular devices. Even very smart researchers who are very careful in experiments are sometimes unable to create new structures, while particularly focused researchers are often quite successful, even if they are not refined researchers. What is the difference? I don’t know.”
Aida points out that patience is also a must in creating new molecular devices. “We can handle failure many times. In fact, it was not until the fifth attempt in the design of the new graphite nanotube that we finally succeeded in creating it. I was, however, on the verge of losing patience. I think if you have about seven failures in succession, you being to think that your basic idea is flawed. Then, you lose your sense of anticipation, and fail to notice positive signs even if they could be the ones leading to the discovery of new functions. I have face-to-face discussions with our researchers, and I sometimes give up on a research theme if the researcher seems to be unable to cope with any more failures. A little more effort may have led to a successful result, but it cannot be helped, because these new devices are being created by real people.”
At the Responsive Matter Chemistry and Engineering Research Group, researchers deal in difficult, painstaking work in the creation of new molecular devices with specific functions. “We have produced very exciting results, including a new molecule that exhibits an interesting phenomenon that was never expected. Unfortunately, however, we cannot speak about the phenomenon yet because the results have not been published.” Look for the results to be published by the Responsive Matter Chemistry and Engineering Research Group in the near future.
-------------------------------------------
About the Researcher
Takuzo Aida
Takuzo Aida was born in Oita Prefecture, Japan, in 1956. He received a bachelor degree in physical chemistry from Yokohama National University in 1979, and then earned his PhD in polymer chemistry in 1984 at the Graduate School of Engineering, University of Tokyo. After serving as a researcher and lecturer, he was appointed associate professor of the Department of Chemistry and Biotechnology at the same university in 1991, and was later promoted to professor in 1996. He joined RIKEN in 2007, where he pursues his research interests in macromolecular chemistry, supramolecular chemistry and bio-science-related chemistry.
Figure 2: Graphite nanotube. Graphene molecules bearing hydrophobic and hydrophilic moieties bond together at the hydrophobic moiety, forming molecular pairs that build up in a spiral pattern to creating a tube. The structure of the graphite nanotube was determined by structural analysis at the Structural Materials Science Laboratory of the RIKEN Spring-8 Center.
Figure 3: A pair of light-activated molecular pliers. Exposure to visible light cause the moiety mounted on the grip to contract, clamping the object between the jaws of the pliers. The body can then be twisted. New molecular devices could induce chemical reactions in the object while it is twisted. A video is available on the website.