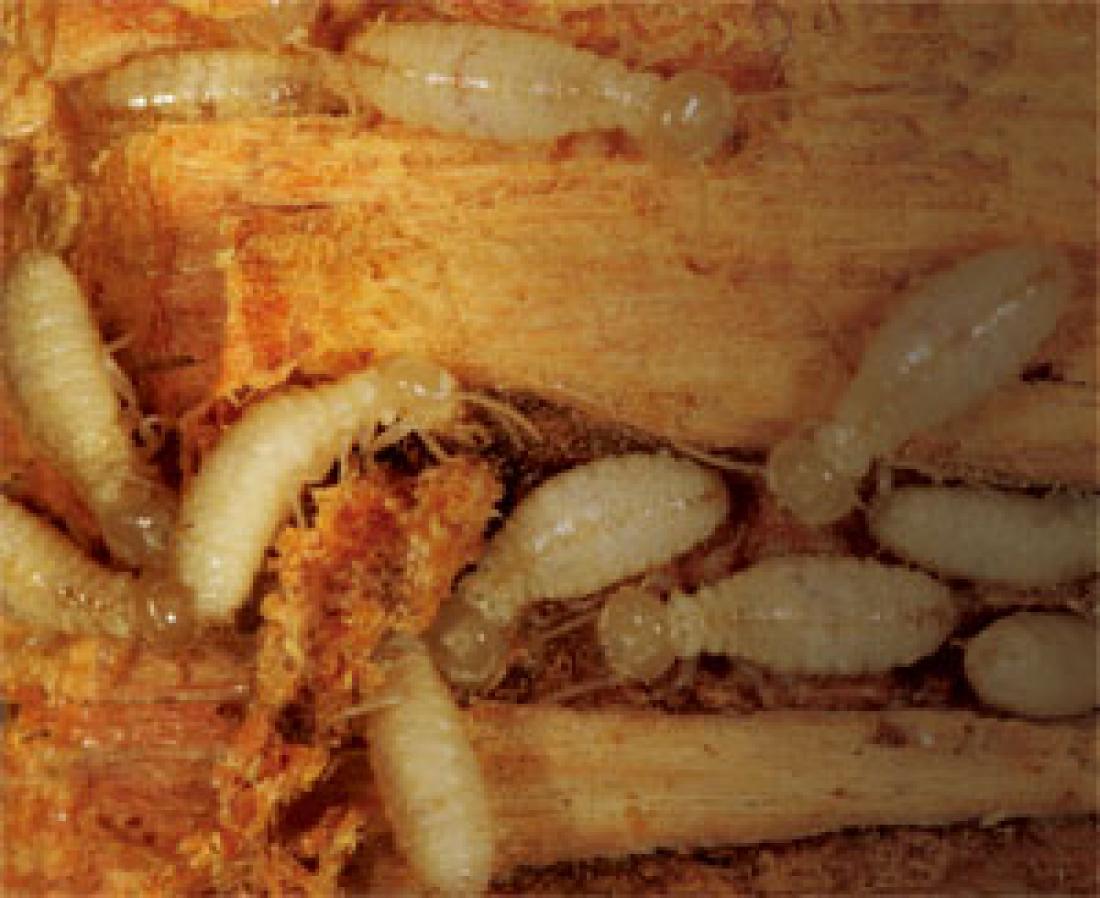
Formosan subterranean termites. Although termites dispose of their gut microorganisms at the time of each ecdysis, they reacquire a full set of microorganisms by nutrient exchange—an interesting process of eating the excreta from the anus of another termite in the nest. It is hypothesized that the social behaviors of termites, including nutrient exchange and community life, have evolved in order to perpetuate the set of gut microorganisms essential for their survival.
Moriya Ohkuma
Head of the Japan Collection of Microorganisms
RIKEN BioResource Center
Investigations are under way all over the world to produce biofuels by degrading the cellulose contained in plant waste, such as wood chips, thinned wood and rice straw, rather than producing biofuels from cereals such as corn and sugarcane. However, as the technology currently available for this purpose consumes a great deal of energy, the efficiency of the production process must be increased. In nature, one organism in particular obtains nutrients by degrading cellulose with extremely high efficiency: termites. These insects exhibit astonishingly high reproduction rates while feeding on wood alone. The key to this efficiency in the production of nutrients is the assemblage of several hundred kinds of symbiotic microorganisms living in the termite gut. However, because most of these microorganisms are unculturable, their identities and natures cannot be examined in detail. For this reason, the symbiotic mechanisms between termites and microorganisms remain unclear. In 2008, Moriya Ohkuma, head of the Japan Collection of Microorganisms at the RIKEN BioResource Center, and colleagues succeeded for the first time in completely decoding the genome (the total genetic information of a set of chromosomes) of a symbiotic microorganism in the termite gut. Described below is his genetic analysis of the mechanisms behind the symbiosis between termites and microorganisms.
99% of microorganisms remain uncharacterized
Koki Horikoshi, researcher emeritus and former chief scientist at RIKEN, was the first to discover a microorganism capable of surviving in an alkaline environment. The cellulose-degrading enzyme produced by it is utilized in a household detergent.
“Japan and RIKEN have traditionally been active in research into microorganisms that degrade cellulose. However, there have been no studies at all on gut microorganisms in termites, which are known to degrade cellulose with extremely high efficiency. This is because most gut microorganisms cannot be cultured by any existing technique,” says Ohkuma.
The properties of a microorganism can be examined in detail by isolating and culturing it to increase its population. If cultivation is not possible, even the very existence of the microorganism may not be known. “Microorganisms that can be cultured by currently available techniques account for less than 1% of all microorganisms identified so far. More than 99% are unculturable, so their identities and properties remain unknown. There are still large frontiers in microbial research.”
Gut microorganisms found to have different habitats in the gut
In 1994, Ohkuma joined RIKEN and began studying microorganisms in the termite gut, an entirely new area of research. “My work at RIKEN started with the capture of termites. I attempted to culture the microorganisms I found in their guts, but I was able to culture less than 1% of them.”
Ohkuma then decided to examine the properties of unculturable microorganisms by genetic analysis. The gut microorganisms in termites can be classified into two types by size: major-member protozoans, which have nuclei, and minor-member bacteria, which lack nuclei. Ohkuma first analyzed genes serving as indexes for classification and identification of the microorganisms. “Surprisingly, new bacterial species were discovered that had not been known, even at the level of phylum, the highest taxonomic unit of bacteria.”
Examination of the distributions of the new bacteria in the termite gut revealed their symbiotic association with particular protozoans. “It was found that particular bacterial species occur as symbionts with respective protozoans. Different bacteria have different habitats in the termite gut.”
A world first in decoding a microbial genome from a trace amount of sample without culturing the microorganism
In recent years, many researchers have investigated unculturable microbial populations by a technique known as metagenomic analysis, resulting in major achievements including some related to the study of human enteric bacteria. “Metagenomic analysis, a method of analyzing a mixture of genes of various microorganisms, is a valuable tool for clarifying the functions and other features of a microbial population as a whole and thereby discovering useful genes. However, the symbiotic mechanisms involved cannot be explored without knowing the capabilities and interactions of individual microorganisms. Metagenomic analysis does not provide information on which genes are present and which ones are lacking in a particular microorganism,” says Ohkuma.
In response, Ohkuma began studying ways of decoding the genome of a microorganism using very few microbial cells and without culturing the microorganism, a goal that no-one in the world had yet achieved. The genome cannot be decoded unless amplified to increase its quantity. If the number of cells of a microorganism can be increased by culturing it, a sufficient amount of the genome can be obtained to allow it to be decoded. In decoding the genome of an unculturable microorganism, the key issue is how to obtain the required amount of the genome.
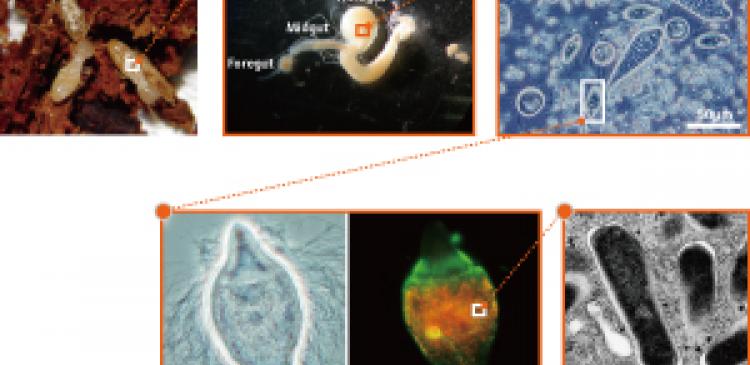
Ohkuma and his colleagues chose Rs-D17 as the first target of their project to decode microbial genomes. This is a newly discovered bacterial species occurring as a symbiont in the gut of the Japanese subterranean termite (Reticuliformes aperatus), for which no strain in culture had been available even at the phylum level. The microorganism lives in the bodies of certain protozoans that degrade cellulose, and accounts for 4% of all symbiotic bacterial cells in the gut of the Japanese subterranean termite. However, the genomic information of Rs-D17 varies slightly depending on its protozoan host, and the presence of this dissimilar genomic information interferes with the decoding. Studies on how to decode the genome from a single bacterial cell have yet to be successful because the amount of genome extractable from a single cell is at present insufficient for analysis.
“Fortunately, Rs-D17 bacterial cells having exactly the same genomic information congregate densely at a particular site in one protozoan. We recovered several hundred Rs-D17 cells from that site. Even so, the amount of genome obtained was extremely low, just one picogram. Decoding the genome requires more than one microgram of genome sample, one million times the amount we had available. Using a new enzyme that allows genome amplification, we amplified the genome ten million times, giving us ten micrograms, and attempted to decode the genome in cooperation with the RIKEN Yokohama Institute. We were thus able to decode the genome of a microorganism completely from very few microbial cells without culturing the microorganism for the first time in the world.”
Symbiotic mechanisms clarified in part
What has decoding the Rs-D17 genome revealed? The genome was estimated to comprise 761 genes for protein synthesis. Also discovered were 121 pseudogenes that had lost their original function for protein synthesis. Another important finding was that the genome is very small, only about a quarter of the size of the Escherichia coli genome. “It is hypothesized that evolution for genome shrinkage occurs, by which genes no longer necessary for Rs-D17 to survive in protozoans lose their function and are discarded. For example, a gene involved in the synthesis of the cell wall, which protects the cell body against environmental changes, has lost its function in Rs-D17. Living in the protozoan body, Rs-D17 does not require genes for environmental adaptation.”
Conversely, still-functioning genes are thought to be indispensable for symbiosis with the termites and protozoans. Discovered among the 761 genes were genes for the synthesis of vitamins and amino acids that cannot be produced by termites or protozoans.
Ohkuma and his colleagues also succeeded in decoding the genome of CfPt1-2, a bacterium that occurs as a symbiont with a cellulose-degrading protozoan in the gut of the Formosan subterranean termite (Coptotermes formosanus). Accounting for 70% of the total cell count of symbiotic bacteria in the termite gut, CfPt1-2 was found to have a gene for the synthesis of nitrogen components, which are scarce in wood fiber. Nitrogen is an essential element for the survival of organisms. CfPt1-2 fixes atmospheric nitrogen and synthesizes ammonia, which serves as a starting material for vitamins and amino acids. It also incorporates ammonia and urea as nitrogen waste from the host protozoan and reuses them as sources of nitrogen. It is believed that by doing so, the termite and protozoan are able to feed exclusively on wood fiber without suffering a shortage of nitrogen.
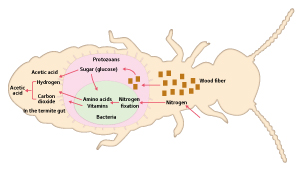
“Termites eat wood fiber, and protozoans degrade the cellulose component into sugars such as glucose, which gets supplied to the bacteria. The bacteria fix atmospheric nitrogen and synthesize amino acids and vitamins using the sugars as a source of energy, and supply the products to the termites and protozoans. There is a firmly established association in a waste-free give-and-take relationship; the termites, protozoans and bacteria are mutually indispensable partners. Part of these remarkable symbiotic mechanisms of extremely high efficiency was clarified for the first time by our bacterial genomic analysis,” says Ohkuma.
“Our next target is the genomes of protozoans that degrade cellulose to sugars,” he says. However, the protozoan genomes are more than 100 times larger than the bacterial genomes. Such large genomes cannot be amplified and decoded using existing techniques. Hence, Ohkuma and others are conducting research on how to extract individual genes expressed in a protozoan population and examine their functions, rather than pursuing genomic analysis. “We don’t know which enzymes are degrading cellulose to sugars in a single protozoan. I want to find that out. Additionally, protozoans have been found to possess a strong ability to produce hydrogen. As for bacteria, Rs-D17 has also been shown to be a hydrogen producer, and hydrogen is likely to be used as a next-generation source of energy.”
Utilizing the symbiotic mechanisms in microorganisms
Biofuels manufactured from cereal crops such as corn and sugarcane are attracting attention as a next-generation energy source. However, the production of biofuels from cereals is causing food prices to surge, and there is increasing deforestation as producers expand agricultural land. Accordingly, there are many investigations focusing on the generation of biofuels from cellulose, which is available in large quantities in plant waste, such as wood chips and rice straw. Provided that cellulose is degraded into sugars, it should be possible to produce fuel alcohol from the sugars using the conventional technology for making Japanese sake. However, the currently available techniques consume vast amounts of energy to achieve the rigorous processing conditions for cellulose degradation, including high pressure and high temperature. “That approach seems to be of no use in resolving the energy crisis. Termites degrade and use cellulose much more efficiently. If we can use the symbiotic mechanisms in microorganisms that allow their host termites to achieve this performance, it will be possible to manufacture biofuels and hydrogen by using the mechanisms artificially to degrade cellulose to sugars with even higher efficiency than termites.
“Humans are using only a part of the capabilities of microorganisms,” continues Ohkuma. “Currently, only single microorganisms and some enzymes produced by microorganisms are being used in practice. Although combinations of two or more microorganisms are used for some purposes, including wastewater purification, the mechanisms involved in their interactions remain unknown. Like the gut microorganisms in termites, microorganisms with a wide range of different capabilities gather and interact with each other everywhere in nature, and build symbiotic systems of extremely high efficiency. If we can understand the mechanisms involved, we might be able to make better use of the excellent capabilities of microorganisms.”
Making best use of information on unculturable microorganisms to compile bioresources
After working at the RIKEN Advanced Science Institute, Ohkuma became head of the Japan Collection of Microorganisms (JCM) at the RIKEN BioResource Center in April this year. The term ‘bioresources’ refers to the types of experimental materials (biological resources) that are indispensable in life sciences research, including animals, plants, microorganisms, cells and genes. Japan is promoting the National Bioresources Project, which aims to establish world-class fundamental infrastructure for bioresources by 2010. The JCM is serving as the hub for activities related to general microorganisms through the project.
“In recent years, there has been a dramatic increase in interest in microorganisms. This is because close mutual associations have been demonstrated between microorganisms and humans, other animals and plants. For example, microorganisms living in the human intestines and skin are closely associated with human health and immune functions. Plant productivity also relies on symbiotic microorganisms that occur around the roots of plants. Utilization of microorganisms is indispensable for resolving energy and environmental issues. In the US and Europe, researchers across a broad range of fields are partnering with microbiologists in large research projects. Unfortunately, Japan is no longer as active in microbial research as it used to be. The same is true at RIKEN, which now has fewer laboratories studying microorganisms. I am very concerned about the situation.”
Ohkuma points out that in view of the situation, the JCM should play a major role. “I want to provide microbial bioresources for researchers working in diverse fields, including medicine, agriculture, energy and the environment, so as to create a new circle of cooperation in microbial research.
Ohkuma also wishes to develop a new type of bioresource. “Currently, only culturable microorganisms are preserved and made available by the JCM. Of course, such microorganisms will remain important and we will continue to expand their stock. But the scope of cooperation should be expanded if we are to develop, preserve and supply genetic information on bioresources from microorganisms that are currently unculturable. I want to make best use of our technical resources that have been nurtured through termite research in the development of new bioresources.” Cooperative research based on new bioresources will open up the vast potential of microorganisms.
About the Researcher
Moriya Ohkuma
Moriya Ohkuma was born in Saitama, Japan, in 1964. He graduated from the Department of Agricultural Chemistry of the University of Tokyo in 1988, and obtained his PhD in 1993 from the same university. He was a Japan Society for the Promotion of Science special fellow at the University of Tokyo until 1994. He then moved to RIKEN as a research scientist and started his career in molecular microbial ecology. He was promoted to senior scientist in 2000, served as a team leader in two research groups at RIKEN, and is now head of the Japan Collection of Microorganisms at the RIKEN Bioresource Center. From 2002 to 2005, he served concurrently in the Japan Science and Technology Agency PRESTO program. His research background is in molecular biology, microbiology, microbial ecology and molecular phylogeny, and his current research interests include symbioses, microbial diversity and microbial functions.
Symbiotic mechanisms in gut microorganisms in the termite intestine. Fractions of wood eaten by termites are taken by protozoans, which decompose the cellulose component of the wood into sugar (glucose) and supply it to bacteria. The bacteria fix atmospheric nitrogen and synthesize amino acids and vitamins using the sugar as a source of energy, and supply the products to the termites and protozoans. The bacteria also produce acetic acid and hydrogen from the sugar; the protozoans produce acetic acid, hydrogen and carbon dioxide from the sugar. Other bacteria in the termite gut produce acetic acid from the hydrogen and carbon dioxide. The acetic acid thus produced serves as a source of energy for the termites.