RIKEN’s old accelerator, which had been barely holding on to life, was brought back to full operation by the nano-beam project (from right: Walter Meissl, Naoko Imamoto, Yasunori Yamazaki).
From a nucleus to mitochondria, lysosomes and the nuclear pore complex, every animal cell contains a range of organelles within just 1–100 micrometers of space. How might cell functions change if one of these organelles becomes damaged? Despite rapid progress in molecular biology research, such experiments have yet to be fully developed because organelles are too small and fragile to be manipulated individually.
Physicist Walter Meissl has been trying to solve this problem by developing an ultra-narrow ion beam that can pinpoint a single organelle while leaving the surrounding cellular functions intact. Since joining RIKEN in March 2009, the Austrian postdoctoral fellow has succeeded in hitting a nucleus with the ‘nano-beam’, and is now preparing for his ultimate target: a centrosome, one of the smallest organelles. “If a nucleus was like a soccer ball, a centrosome would be only one little point on that ball,” Meissl says.
Creating nano-beams using glass capillaries
Meissl is a central figure of the nano-beam project, which was originally established with a grant from the President’s Fund for fiscal 2007 and 2008. The project was initiated by Yasunori Yamazaki, chief scientist of the Atomic Physics Laboratory at the RIKEN Advanced Science Institute, in collaboration with Naoko Imamoto, chief scientist of the Cellular Dynamics Laboratory at the same institute. Although the grant ended in March 2009, the researchers still continue to work together in an effort to implement what they call ‘cell surgery’.
Previously from the Vienna University of Technology in Austria, Meissl first heard about the project when Yamazaki gave a talk at his institute in 2008. “I was intrigued with the idea of getting into the interdisciplinary work between physics and biology,” says Meissl, who then decided to join Yamazaki’s laboratory.
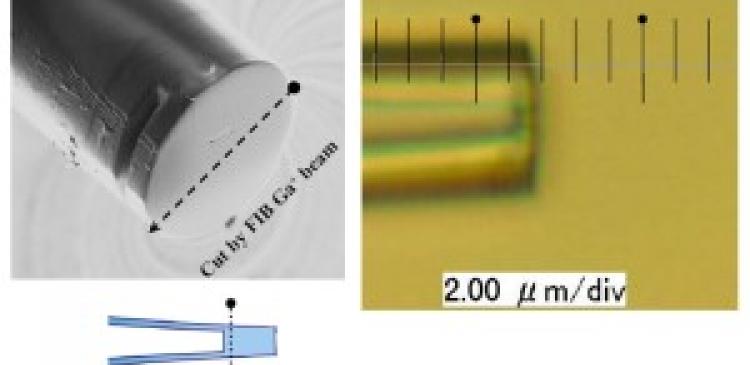
Meissl’s work is a small but highly innovative product of Yamazaki’s lab, the main focus of which is the investigation of exotic collision products such as antihydrogen atoms and the development of advanced cooling techniques to capture these particles. The lab has also been developing slow, highly charged ion beams (the aggregation of charged atoms and molecules) with nanometer-scale diameters using glass capillaries. Yamazaki says it is extremely difficult to focus highly charged ions into nano-sized spots, and other groups around the world have been attempting to do so using dedicated lenses that combine the effects of electric and magnetic fields. No-one had thought of using glass, because it does not conduct electricity and is susceptible to the build-up of static electricity, which deteriorates the quality of the ion beam, he says.
Instead, Yamazaki took advantage of glass’s insulation properties. When ions are first injected into the inlet of a glass capillary, they accumulate on the capillary’s inner wall; when the accumulation of ion charge on the inner walls becomes sufficient, subsequently injected ions are naturally guided all the way to the outlet. At a cost of 50 yen (US$0.40) per capillary, “it was so simple, like a joke, but we could confirm the beams were strong enough,” says Yamazaki.
A tweak for biological applications
Nano-beams can be used to manipulate molecules and atoms on surfaces, so demand is growing for their use in the fabrication of semiconductor materials. But Yamazaki wanted to use the technology for unconventional purposes, and sought ideas at one of RIKEN’s informal chief scientist meetings, at which chief scientists with various backgrounds come together to learn about each other’s activities. Yamazaki became intrigued with the potential biological applications of his nano-beams, and suggested a collaboration with Imamoto, whose primary area of study is in the regulation and maintenance of nuclear function. “But at first, my idea was rebuffed,” Yamazaki says, “because the beams can only be produced in a vacuum chamber, and cells die without air.”
Yet Yamazaki was undaunted and hit upon the idea of adding a thin glass cap at the capillary outlet so it can be immersed in a liquid while maintaining the capillary vacuum1, 2. The beam can be controlled so that it travels only 100 nanometers to several micrometers and the ions have enough energy to penetrate the window, allowing it to be used to irradiate a single organelle, or even a part of one, Yamazaki says. Another advantage of glass is that it is transparent and thus enables researchers to observe the irradiation point directly using an optical microscope. Compared to conventional ionizing radiation, which does not have the precision or selectivity of the nano-beam, "the new beam could lead us to observe more precisely how the damage to an organelle affects overall cell dynamics,” Imamoto says.
A physicist learns how to culture cells
Yoshio Iwai, a postdoctoral researcher who recently left the laboratory, spent the first 18 months of the project constructing a dedicated beam line for a small tandem accelerator at RIKEN. Now, much of the work has been handed over to Meissl. Unlikely for a physicist, he started with learning from Imamoto how to culture a cell, multiply it, and prepare solutions for experiments. “It is very exciting and totally different from my previous work because a living cell shows unpredictable results, a drastic change from surface physics,” Meissl says.
Meissl also made some additional changes to the beamlines. “Usually in physics, you fight for more ions, more intensity. But for biological experiments, we need as little radiation as possible because even a single particle can harm a cell.” He installed a fast beam switch that allows attenuation of the ion beam down to short packets containing as little as a single ion.
From nucleus to centrosome
The most difficult part of the experiments is manually setting up the cell in the best position for nano-beam irradiation, Meissl says. An equally important step is to optimize the strength of the ion beam and calculate the right irradiation time. By switching the beam on and off in less than one microsecond, Meissl takes multiple shots at the cell surface, but each time he obtains different results. “I need much more target practice,” he says. “It’s not as easy an experiment as it looks. A lot of patience is required,” Imamoto adds.
In the summer of 2009, Meissl succeeded in hitting a nucleus, and the cell died immediately. After a number of attempts, he has fine-tuned the strength of the beam and is now able to hit the nucleus while keeping the cell alive. He is now preparing to target a centrosome. At less than one micrometer in diameter, the centrosome exists as a pair of organelles floating near the nucleus, and organizes microtubules to divide chromosomes into daughter cells during cell division. Yamazaki and Imamoto are curious to see what will happen if one of the pair is damaged, but Meissl says it is incomparably more difficult than targeting a nucleus.
“The project is just becoming science,” Yamazaki says. “We have just begun to explore the potential of this new technique that can lead to unprecedented applications bridging biology and physics.”