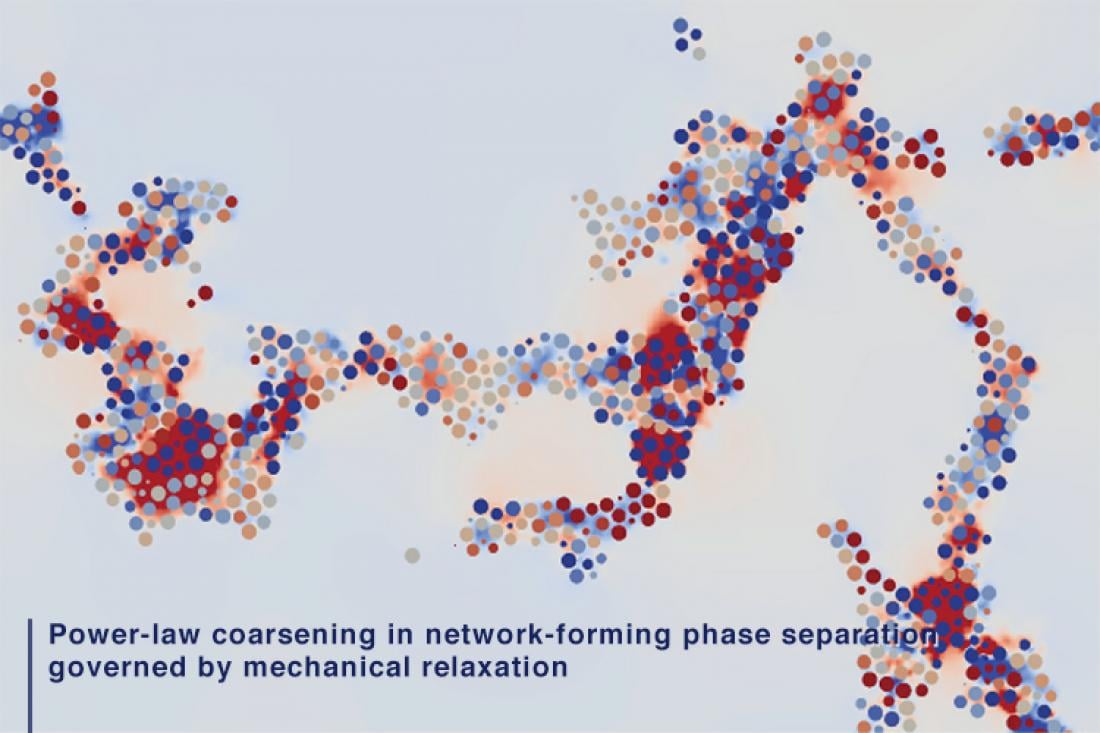
Tokyo, Japan – Researchers from Institute of Industrial Science at The University of Tokyo investigated the mechanism of phase separation into the two phases with very different particle mobilities using computer simulations. They found that slow dynamics of complex connected networks control the rate of demixing, which can assist in the design of new functional porous materials, like lithium-ion batteries.
According to the old adage, oil and water don’t mix. If you try to do it anyway, you will see the fascinating process of phase separation, in which the two immiscible liquids spontaneously “demix.” In this case, the minority phase always forms droplets. Contrary to this, the researchers found that if one phase has much slower dynamics than the other phase, even the minority phase form complex networks instead of droplets. For example, in phase separation of colloidal suspensions (or protein solutions), the colloid-rich (or protein-rich) phase with slow dynamics forms a space-spanning network structure. The network structure thickens and coarsens with time while having the remarkable property of looking similar over a range of length scales, so the individual parts resemble the whole.
In the case of spontaneous demixing, the self-similar property causes the typical size of the domains to increase as a function of the elapsed time while obeying a power law. Classical theories predict that the growth exponent of the domains should be 1/3 and 1 for droplet or bicontinuous structures, respectively. However, for network-forming phase separation, it has not been explored how the structure grows or if there is such a law.
Now, using large-scale computer simulations, researchers at The University of Tokyo studied how the typical size of phase domains grows over time when a system is deeply quenched. “In such a situation, the particle mobility can be significantly different between the two phases, and then, the classical theory does not necessarily apply, ” first author Michio Tateno says. The team studied the phase separation of a fluid into a gas and liquid and the demixing of a colloidal suspension consisting of insoluble particles and a liquid, using molecular dynamics simulations and hydrodynamic calculations, respectively. They found that the minority phase of slow dynamics universally forms a network structure that grows with a growth exponent of 1/2, and provided a theoretical explanation for the mechanism.
“Significant differences in the particle mobility between the two phases plays a critical role in controlling the speed of the demixing process,” senior author Hajime Tanaka says. Because many devices, like rechargeable batteries and catalysts, rely on the creation of intricate porous networks, this research may lead to advances in these areas. In addition, it may shed light on certain cellular functions that have been hypothesized to be controlled by internal biological phase separations.
###
About Institute of Industrial Science (IIS), the University of Tokyo
Institute of Industrial Science (IIS), the University of Tokyo is one of the largest university-attached research institutes in Japan.
More than 120 research laboratories, each headed by a faculty member, comprise IIS, with more than 1,000 members including approximately 300 staff and 700 students actively engaged in education and research. Our activities cover almost all the areas of engineering disciplines. Since its foundation in 1949, IIS has worked to bridge the huge gaps that exist between academic disciplines and realworld applications.