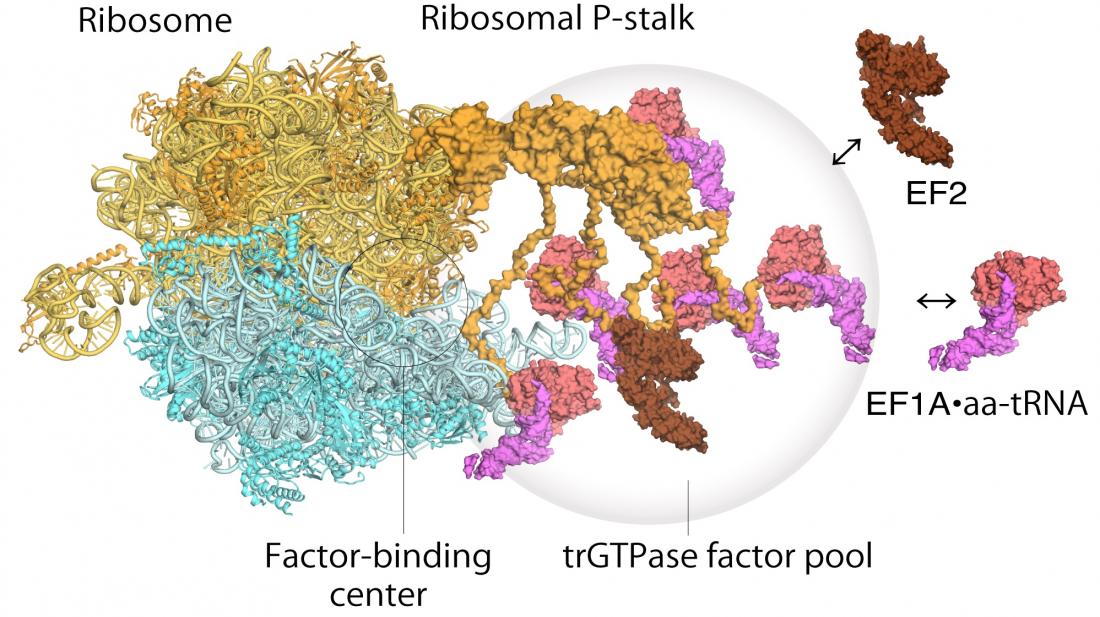
Image: Model of translating ribosomes and elongation factors. EF1A•GTP•aatRNA and EF2 assemble to the ribosomal stalk on the translating ribosome. The translation factor pool contributes to efficient protein synthesis in a crowded intracellular environment.
Ribosomes are the complexes of ribonucleoproteins at the heart of protein synthesis in cells. However in the absence of conclusive evidence, how these complexes operate has been open to debate. Now Hirotatsu Imai and Noriyuki Kodera at Kanazawa University, alongside Toshio Uchiumi at Niigata University in Japan, show visualizations of the structural dynamics and factor pooling that take place at ribosome stalk proteins as they build new proteins.
Ribosomes were first discovered in the 1950s and their broad function has been widely understood for some time – they read messenger RNA sequences and from that generate sequences of correctly ordered amino acids into new proteins. The ribosome stalk protein in particular plays an integral role in the protein synthesis process by recruiting protein factors responsible for translation and elongation of the amino acid sequence. However it has been hard to satisfactorily establish the structure of the bound ribosome stalk protein because of its flexibility. Here the high resolution and fast image capture of high-speed atomic force microscopy proved invaluable.
Atomic force microscopy uses a nanoscale tip to feel samples, much like a vinyl record player needle scanning over a record, except that the details identified by an atomic force microscope can have atomic-scale resolution. The versatility of the technique for different surfaces was already a huge advantage for biological studies, but with the advent of high-speed atomic force microscopy the technique was able to capture dynamic processes for the first time as well. Imai, Uchiumi and Kodera used the technique to reveal that the stalk protein actually flips between two conformations – one that agrees with previous structural models and one entirely unexpected new conformation.
As for how the ribosome operates, a two step mechanism had been previously proposed to describe how genetic information is translated through proteins known as “translational GTPase factors”. The first step is the recruitment of the factors to the factor-tethering site on the protein stalk, thereby increasing the concentration of factors there – so-called factor pooling. The second step is the binding and stabilizing of a translational GTPase on the ribosomal factor-binding center to catalyse GTPase hydrolysis. From their high speed atomic force microscopy study the researchers were able to obtain the first visual evidence for the translational GTPase factor pooling mechanism by the ribosomal stalk.
Although the study was unable to give conclusive evidence of the action of the factors once bound, the researchers did note that the factors appeared to be retained in the vicinity once GTPase hydrolysis was complete, suggesting a potential role of the stalk protein in further stages of protein synthesis. The researchers conclude, “future work with HS-AFM will provide further important information to understand the dynamic behaviors of these complex translational machineries.”
[Background]
High-speed atomic force microscopy
Atomic force microscopy was developed in the 1980s and brought the atomic scale resolution achieved by scanning tunnelling microscopy (which won the 1986 Nobel Prize for Physics) to non-conducting samples. It works using a tiny cantilever with a nanoscale tip at the end, which feels the surface. The interactions between tip and surface provide a signal that can be used to generate an image of not just the topography but also in some cases an indication of the chemical composition of structural features in a sample.
While the non-conducting surface imaging of atomic force microscopy brought huge benefits to biological research, these studies were able to move up a gear again when Toshio Ando and his team at Kanazawa University reported an atomic force microscope that operates at high speed. Atomic scale resolution images became movies bringing not just structures but also dynamics within grasp.
Ribosomes
Ribosomes are the protein factories found in all living cells. The genetic DNA sequence that encodes for a series of amino acids in the protein is transcribed to messenger RNA, to which the ribosome binds. The correct amino acids for the protein under construction are then fed to the ribosome by means of transfer RNA.
Translational GTPase factors take on the task of translation and elongation of the amino acid sequence for the protein under construction, making their role critical to the process. The mechanism by which they work with the ribosome in this operation has now been visualized by Imai, Uchiumi and Kodera using high-speed atomic force microscopy.