Figure 1. Principle of new therapeutic treatment
[Background]
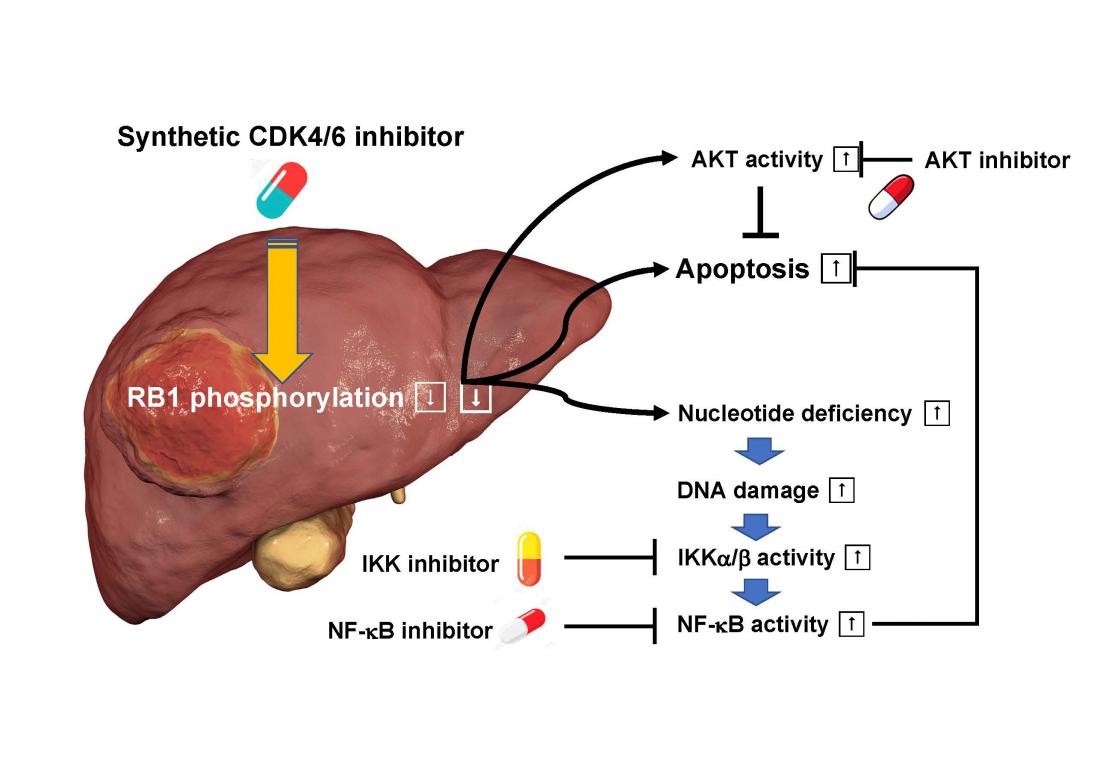
The RB1 tumor suppressor gene*1) protein antagonizes uncontrolled growth of cancer by suppressing cell cycle progression but its function is repressed in most cancers. CDK4/6*2) represses this function by phosphorylating RB1. In contrast, drugs that inhibit CDK4/6 inhibit the phosphorylation of RB1 and restore the cancer-suppressing function of RB1. However, since it was difficult to achieve a sufficient therapeutic effect by a single administration of a synthetic CDK4/6 inhibitor, development of a combination therapy that maximizes the function of the drug has been awaited.
[Results]
The research team led by Prof. Chiaki Takahashi of the Cancer Research Institute, Kanazawa University, in collaboration with a research team led by Associate Prof. Itsuki Ajioka of Tokyo Medical Dental University and with scientists from several domestic and overseas institutions found that hepatocellular carcinoma is generated due to loss of RB1 function in mouse hepatocytes. Then, they established cell lines such as hepatocellular carcinoma into which a mutation that mimics a state in which RB1 is constantly activated was introduced, and performed high-throughput screening in order to find a compound that enhances the effect of a synthetic CDK4/6 inhibitor. As a result, they found that an IKKβ inhibitor shows such an effect. This weakens the function of IKKβ kinase and allows cells to escape from cell death. It was found that when treated with a synthetic CDK4/6 inhibitor alone, IKKα/β and its downstream NF-κB, which detect abnormalities triggered by nucleic acid synthesis deficiency , were activated to avoid cell death, causing insufficient therapeutic effects. Next, they performed similar analyses for K-Ras oncogene*3) mutated lung cancer and colorectal cancer having intact RB1 and found that combined use of another drug that inhibits another kinase, AKT, is effective in a way similar to that obtained by combined use of an IKKβ inhibitor.
[Future prospects]
The synthetic CDK4/6 inhibitors are already covered by health insurance in Japan and the IKKβ inhibitor and the AKT inhibitor are also in clinical trials. Therefore, it is possible to start clinical trials using these drugs in combination with a synthetic CDK4/6 inhibitor. Based on the results of the present study, it is expected that treatments for hepatocellular carcinoma, K-Ras oncogene mutated lung cancer, colon cancer, pancreatic cancer, cholangiocarcinoma, and many other RB1 wild-type refractory cancers will be improved. Currently, we are calling for various clinical departments to participate in cancer therapy trials.
[Glossary]
*1) RB1 tumor suppressor gene
Germline or sporadic abnormality in the RB1 gene causes cancer such as retinoblastoma. Under normal conditions, the RB1 tumor suppressor gene suppresses uncontrolled growth of cancer by suppressing the expression of cell cycle genes. The RB1 tumor suppressor gene product is inactivated in a reversible manner in various cancers.
*2) CDK4/6
Cyclin-dependent kinases 4 and 6 are collectively referred to as CDK4/6. When cyclin D is upregulated by cell proliferation signaling and these kinases are activated, RB1 is phosphorylated but only at one site, i.e. monophosphorylated. After a while, the cyclin E-CDK2 complex inactivates RB1 completely by subsequent phosphorylation at up to further 13 sites. CDK4/6 inhibitors stress cancer cells by leaving RB1 in a non-phosphorylated state for extended periods of time.
*3) K-Ras oncogene
The most representative oncogene that causes various cancers such as pancreatic cancer, colon cancer, and lung cancer. When this gene product is activated by mutation, the function of RB1 is strongly repressed. Hence, the presence of K-Ras mutation does not require additional mutation or deletion in the RB1 gene.