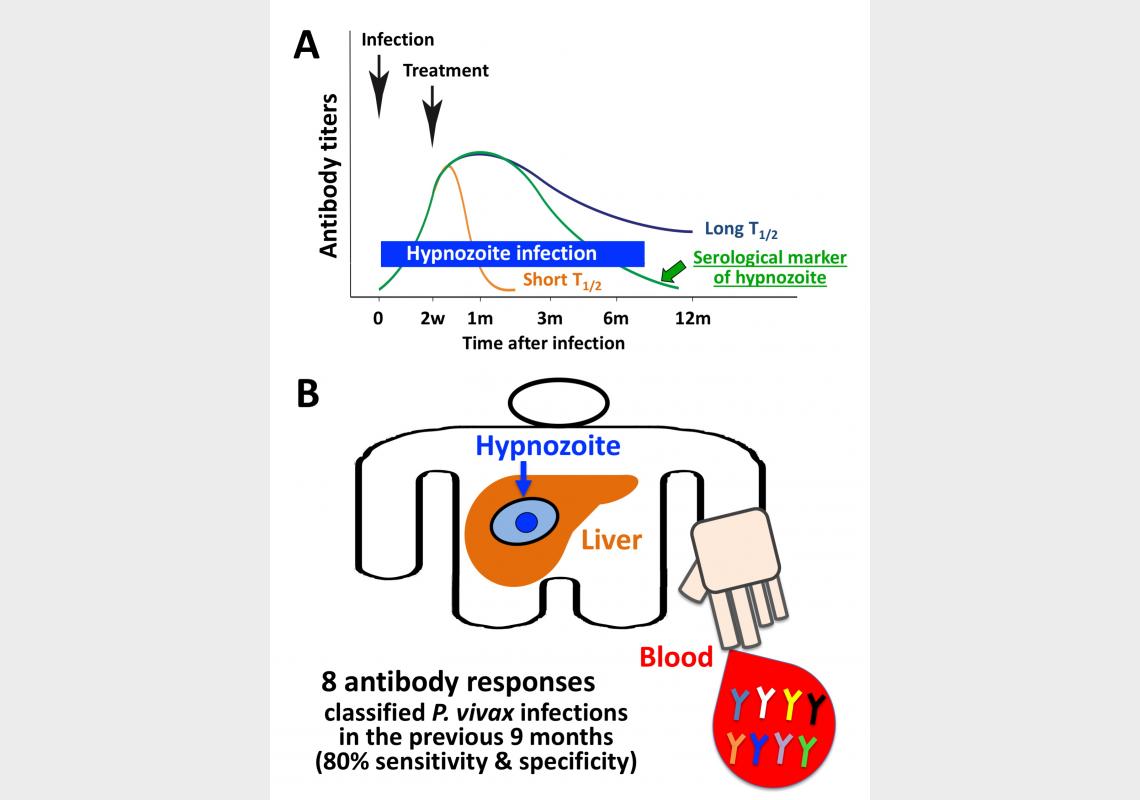
(A) How to detect dormant hypnozoite of P. vivax? Antibodies can predict recent infections if they have the correct kinetic profile (Green line, serological marker of hypnozoite). Antigens selected based on the kinetic profiles in 9 months following clearance of infection are candidates for detecting hypnozoites.
(B) Hypnozoite biomarkers. Eight antibody responses classified P. vivax infections in the previous 9 months with 80% sensitivity & specificity.
Plasmodium vivax is the most widespread malaria parasite worldwide, with up to two billion people at risk of infection. As well as causing illness and death in its ‘active’ stage of infection, the parasite can hide as hypnozoites, a dormant stage, in the liver, and is a significant cause of ‘relapsing’ malaria.
These hypnozoites, undetectable with current diagnostics, can be responsible for >80% of all blood-stage infections. Identifying and targeting individuals with hypnozoites is thus essential for accelerating and achieving malaria elimination. A major gap in the P. vivax elimination toolkit is the identification of individuals carrying clinically silent and undetectable hypnozoites.
This study developed a panel of serological exposure markers capable of classifying individuals with P. vivax infections within the previous 9 months who have a high likelihood of harboring hypnozoites. 1) We measured IgG antibody responses with the AlphaScreen system to 342 P. vivax proteins expressed by a wheat germ cell-free system, invented at Ehime University, in longitudinal clinical cohorts conducted in Thailand and Brazil and identified 60 candidate serological markers of exposure. 2) Candidate markers were validated using samples from year-long observational cohorts conducted in Thailand, Brazil and the Solomon Islands and antibody responses to eight P. vivax proteins classified P. vivax infections in the previous 9 months with 80% sensitivity and specificity. Mathematical models demonstrate that a serological testing and treatment strategy could reduce P. vivax prevalence by 59–69%.
These eight antibody responses can serve as a biomarker, identifying individuals who should be targeted with anti-hypnozoite therapy. The test offers new opportunities for improving malaria control and elimination strategies.