Featured in Asia Research News 2020 Magazine
Her most recent research has focused on chronic myeloid leukaemia (CML), a cancer that develops in blood-forming cells in the bone marrow. Genetic changes in these cells lead to the formation of an abnormal gene, BCR-ABL1, contributing to their transformation into cancer cells. While slow growing, CML can become acute and difficult to treat.
Kuan is taking a closer look at the BCR-ABL1 gene to see if it could be used as a reliable screening tool for CML before it develops. We talked with Kuan about her research and what motivates her.
Q. How did you become interested in studying medicine, and haematology in particular?
A. I had a skin infection on my right lower leg when I was very young. My neighbour offered a topical powder for the infection but it got worse: there was pus, it looked ugly, and my whole leg became swollen. It was the first time for my young heart to feel that life can just fade away. A very nice doctor, Dr Lu Qi Song, treated me and I recovered. The honourable work of the doctor led me to study medicine.
I chose haematology as my subspecialty because I was inspired by the amazing and endless passion of three haematologists towards their patients: Dr Visalachy Purushothaman (1947- 2019), Dato Dr Chang Kian Meng and Dr Jameela Sathar. They showed me the kind of doctor I want to be.
Q. What led you to study CML?
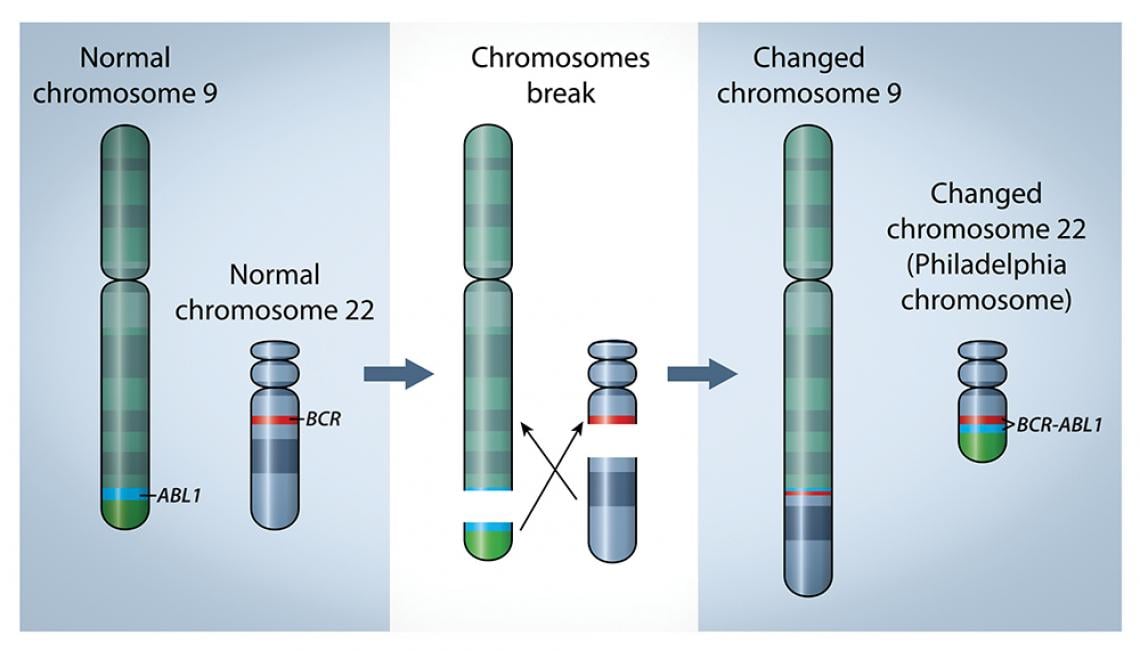
A. CML is the perfect disease to start with. CML has been the leading cancer model in oncology. It was the first cancer whose prognosis changed from doom to near-normal life span, simply with oral medication. It was also the first human cancer to have a clearly identified altered chromosome, called the Philadelphia chromosome, named after the city it was first discovered in back in 1960. Subsequent research on the Philadelphia chromosome led to the discovery of the altered gene, BCRABL1, the genetic marker of CML.
Besides clinical and bone marrow features, a doctor must demonstrate the presence of the Philadelphia chromosome and/or BCR-ABL1 in order to provide a CML diagnosis. The level of BCRABL1 can be easily measured with a blood sample, and the result is standardized all over the world in a unit called International Scale (IS). Other cancers might have measurable markers, but none are as easy or as standardized as CML. Doctors use the IS measurement to determine the magnitude of the disease in CML patients and their response to oral medication.
The genetic marker for chronic myeloid leukaemia is a gene called BRC-ABL1, which forms when two chromosomes break apart and swap sections. The gene is on modified chromosome 22, named Philadelphia chromosome, after the city in which it was discovered.
Q. What do you find most challenging about this research?
A. The most challenging part about this research is how to approach communities. Previous studies had reported that 2% to 57.5% of healthy adults carry the BCR-ABL1 gene. However, the studies did not use community- or population- based methods, so were not appropriate for determining BCR-ABL1 ’s suitability as a screening genetic marker for CML. To conduct a community and population-based study, we need to go to houses selected by proper population survey methods. We have to knock on the door, explain ourselves and get consent for an interview and to take blood samples. This is really challenging for me. I am a clinician and patients come and talk to me everyday because they want to talk to me. Now, it is the other way around. Luckily, my teammate, Dr. Anselm Su Ting, is a public health specialist and helped a lot in planning our community approach for blood sampling.
Another part that is equally challenging for me is the laboratory work, because I am a clinician. The most unforgettable moment was when the -70°C freezer where I stored the extracted ribonucleic acid (RNA) broke and staff transferred the samples to a -30°C freezer. A temperature of -70°C is required to store durable RNA. -30°C is not good enough. My heart stopped and my mind was stunned. Luckily, we caught it in time and were able to store the RNA in a colleague’s freezer.
Q. What are you most excited about right now?
A. Using our community approach to blood sampling, my collaborators at Kumamoto University and I believe we have evidence that the genetic marker for CML can be used as an early screening tool for the disease, even before a patient begins to show symptoms and the genetic changes associated with the diseases begin to occur. It is important to try to detect CML at its earliest stage for the 4% to 18% of cases that will progress to acute levels in order to reduce cancer-related deaths. This is especially important given how slowly this cancer grows; it can go undetected for a long time.
Q. What are your next steps for this work?
A. We plan to expand our initial study published in the International Journal of Hematology, which included 190 blood samples, to a larger population and do longer follow up to determine if the gene can indeed be used as a reliable indicator of developing CML. It appears that the normal population does not usually carry this gene. We plan to further investigate if those who do have it are likely to develop CML.
Further information
Dr Kuan Jew Win | E-mail: [email protected]
Department of Medicine
Universiti Malaysia Sarawak
Read this story in the Asia Research News 2020 magazine.
The many ways we can tell your research story. Find out more from our Content services page.