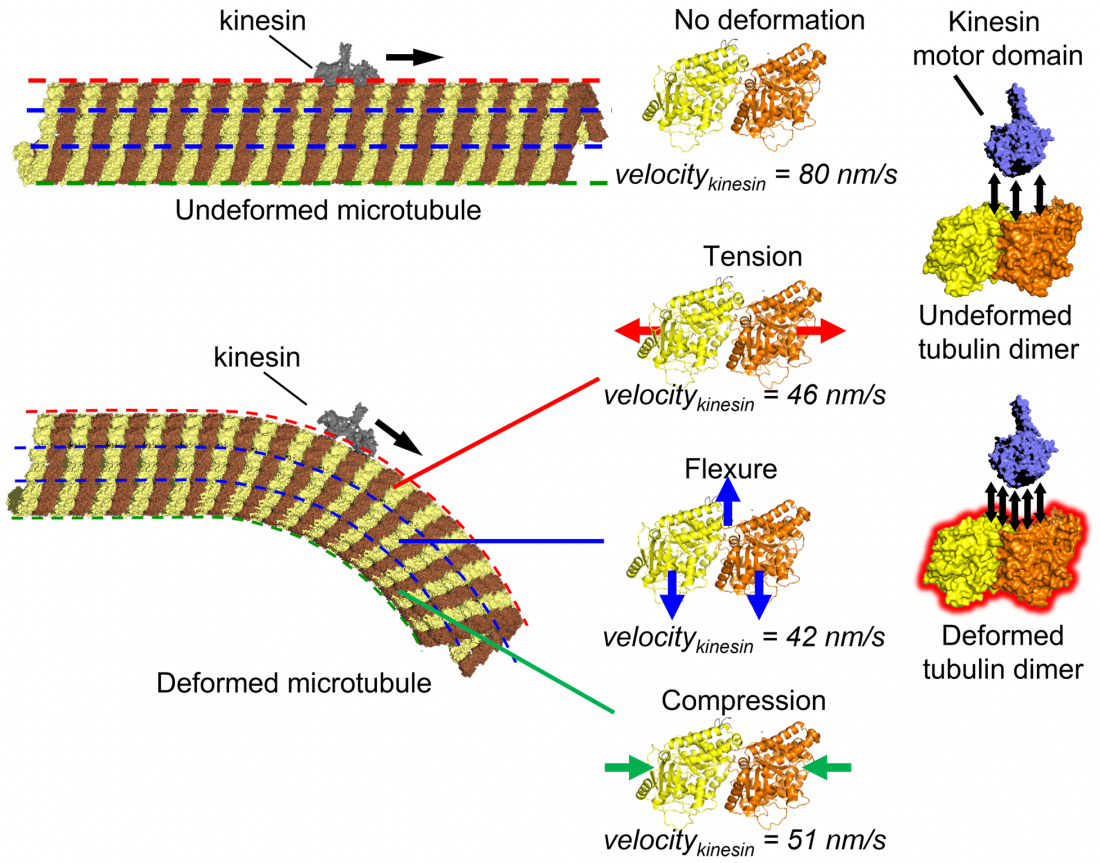
Microtubule, a cytoskeletal filament, may function as a mechano-sensor to regulate intracellular transport by its motor protein, kinesin. Tension, flexure, or compression in the microtubule filament slowed down the kinesin movement along the microtubule due to increased intermolecular interaction, as proved by the molecular dynamics simulation (Syeda Rubaiya Nasrin, Christian Ganser, et al. Science Advances. October 13, 2021).
Joint press release by Hokkaido University; the National Institutes of Natural Sciences; Research Center for Advanced Science and Technology (RCAST), The University of Tokyo; Yokohama City University; and Nagoya University.
Inside cells, microtubules not only serve as a component of the cytoskeleton (cell skeleton) but also play a role in intracellular transport. In intracellular transport, microtubules act as rails for motor proteins such as kinesin and dynein. Microtubules, the most rigid cytoskeletal component, are constantly subjected to various mechanical stresses such as compression, tension, and bending during cellular activities. It has been hypothesized that microtubules also function as mechanosensors that convert mechanical information into biochemical information.
A research team led by Associate Professor Akira Kakugo of Hokkaido University has provided direct evidence that microtubules function as mechanosensors that regulate intracellular transport. The findings were published in the journal Science Advances. The team included Dr. Syeda Rubaiya Nasrin, Seiji Nishikawa, Dr. Arif Md. Rashedul Kabir and Professor Kazuki Sada of Hokkaido University; Dr. Christian Ganser of the National Institutes of Natural Sciences; Associate Professor Takefumi Yamashita of Research Center for Advanced Science and Technology (RCAST), The University of Tokyo; Professor Mitsunori Ikeguchi of Yokohama City University; Professor Takayuki Uchihashi of Nagoya University; and Professor Henry Hess of Columbia University.
Recent studies have shown that mechanical stress has a significant effect on cell differentiation, development, and disease. Some studies have suggested that microtubules, which are the most rigid cytoskeleton and play an important role in cell morphogenesis and intracellular material transport, may also function as mechanosensors; however, no direct evidence has been obtained to support such a speculation.
The high-speed atomic force microscope used in the study (Photo: Akira Kakugo).
The team carried out experiments with microtubules in a cell free system. They found that kinesin movement is slowed down when the microtubule is bent. Using high-speed atomic force microscopy, which allows for the observation of dynamic processes at the atomic level, they confirmed that this phenomenon occurs even without cavities or cracks in the microtubule structure, which were thought to be the main reason behind the slowing. Analysis of the kinesin binding affinity showed that bent microtubules appear stickier to kinesin compared to unbent microtubules. From all-atom molecular dynamics simulations, it was found that the mechanism of such slowing down involves the enhanced interaction energy of kinesin and deformed microtubule structural units.
The results of this research have implications in the fields of biomechanics and mechanobiology, that study the mechanical environment in and around cells; in research on neurological diseases caused by impeded axonal cargo transport; and for the development of mechanical sensors by biomolecular proteins.
From the left: Syeda Rubaiya Nasrin and Akira Kakugo, Hokkaido University; Christian Ganser, National Institutes of Natural Sciences; Takefumi Yamashita, RCAST, The University of Tokyo; Mitsunori Ikeguchi, Yokohama City University; Takayuki Uchihashi, Nagoya University; and Henry Hess, Columbia University (Photos: Syeda Rubaiya Nasrin, Akira Kakugo, Christian Ganser, Takefumi Yamashita, Mitsunori Ikeguchi, Takayuki Uchihashi, Henry Hess).