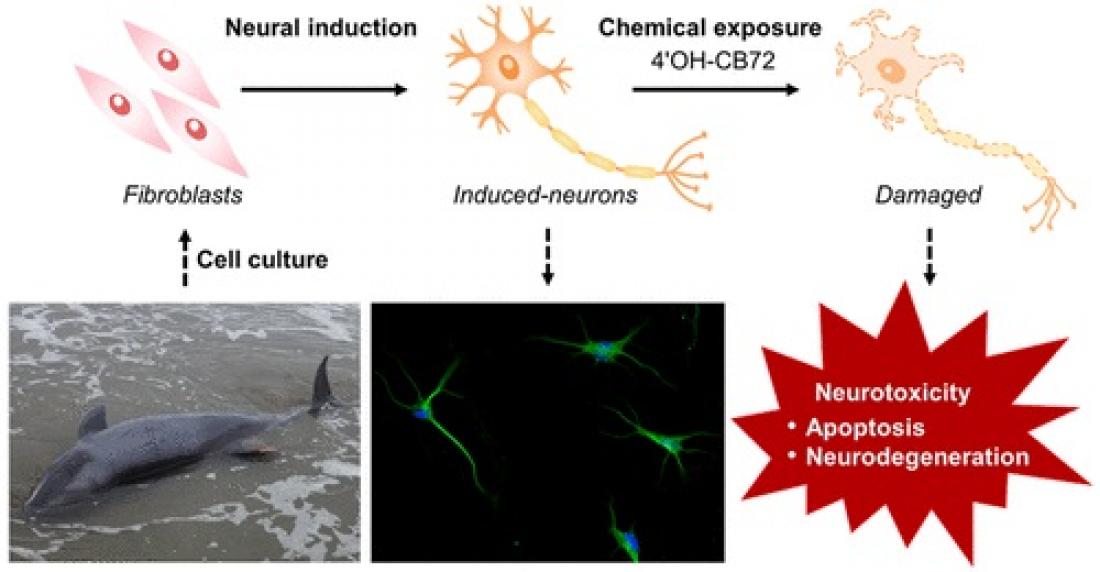
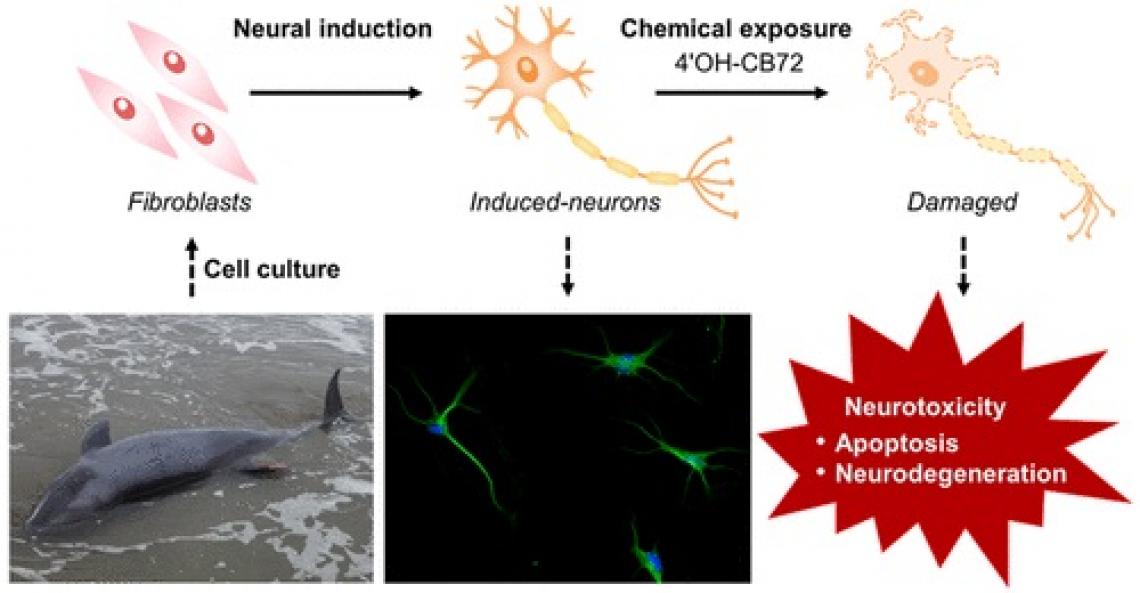
Whales accumulate large burdens of environmental pollutants that threaten their survival and health. Toxicological studies on cetacean species have been extremely challenging because invasive studies are restricted by legal and ethical considerations and sampling of wild cetaceans is highly opportunistic. Although model animal studies can provide data from practical experiments, extrapolating the toxicological effects to cetaceans is limited due to the large interspecies susceptibility to chemical exposure. The types of whale cells that can be cultured are limited, and cell-specific assays for whales have not been developed. A research team of the Center for Marine Environmental Studies (CMES) of Ehime University, Japan succeeded in direct reprogramming the fibroblasts of stranded melon-headed whales (Peponocephala electra) to neurons, not through the induction of pluripotent stem cells (iPSCs), but by using a cocktail of small compounds. Using whale induced neurons, they have investigated the neurotoxicity of an environmental pollutant on cetacean neurons for the first time. Their research was recently published in Environmental Science & Technology.
Reprogramming from fibroblasts to neuronal cells
The research team obtained tissue samples from melon-headed whales mass stranded along the coast of Hokota-city, Ibaraki, Japan. After a few weeks of treatment with a cocktail of small compounds, the morphology of the fibroblast cells derived from the melon-headed whale tissues changed to neuron-like cells. Reprogramming of the fibroblasts to neurons, induced neuronal cells (iNCs), was confirmed by positive signals for neuronal markers: beta-III tubulin (Tuj-1) and microtubule associated protein 2 (MAP2), and negative signals for astrocyte and oligodendrocyte markers: glial fibrillary acidic protein (GFAP) and 2’,3’-cyclic nucleotide 3’-phosphodiesterase (CNPase), respectively. Transcriptome analysis of the whale iNCs also supported the success in reprogramming, showing downregulation of a fibroblast marker elastin (ELN) and upregulation of neuron-related genes such as synaptic genes: synaptophysin (SYP) and stathmin 3 (STMN3).
Apoptosis assay
Apoptosis was measured by detecting nuclear chromatin fragmentation by TUNEL assay. Over 80% of whale iNCs have led to apoptosis after 24hr exposure to 20μM 4’OH-CB72, with apoptosis-positive cells increasing 1.8-2.4 times compared to vehicle controls.
Transcriptome analysis of whale neuronal cells exposed to 4’OH-CB72
Upregulation of the genes related to apoptosis in whale iNCs was also confirmed by transcriptome analysis. Genes such as apoptosis inducing factor mitochondria associated 1 (AIFM1), BH3 interacting domain death agonist (BID), andtumor necrosisfactor (TNF) receptor associated factor 2 (TRAF2) were upregulated in whale iNCs. On the other hand, cell survival-related genes were downregulated in whale iNCs. Additionally, many genes involved in neurodegenerative diseases such as Alzheimer’s disease, amyotrophic lateral sclerosis, and Huntington’s disease were altered in whale iNCs exposed to 4'OH-CB72.
Bioinformatics analyses using differentially expressed genes upon exposure to 4’OH-CB72 suggested that the cellular signaling pathways of mitochondrial dysfunction, chromatin degradation, axonal transport, and ubiquitin−proteasome system were disrupted by this pollutant. These effects ultimately lead to neurodegeneration through neuronal apoptosis and cell death.
Future perspectives
Since cetaceans are chronically exposed to a variety of environmental pollutants, not only 4’OH-CB72, but also other PCBs, the neurotoxicity of other compounds is also of concern. This approach may be useful for other marine mammals for which there is yet no effective means of neurotoxicity testing.
Exposure of a PCB metabolite to whale-derived induced neurons caused apoptosis and neurodegeneration