Electromicroscopic image of West Nile virus colored in yellow. Courtesy of Cynthia Goldsmith, P.E. Rollin, USCDCP.
West Nile virus (WNV) inhibits autophagy — an essential system that digests or removes cellular constituents such as proteins — to induce the aggregation of proteins in infected cells, triggering cell death and brain inflammation (encephalitis), according to Hokkaido University researchers. They also discovered that a drug can induce autophagy to remove protein aggregates and thus prevent cell death.
West Nile fever is a zoonosis spread by the bite of an infected mosquito. West Nile fever outbreaks have been reported across the world, mainly in North America and Europe, and caused hundreds of deaths in the past few decades. After a human becomes infected, virus replication temporarily occurs in peripheral tissues. In some patients, the virus enters the brain, infecting neural cells and causing cell death and serious cases of encephalitis.
The team previously found that WNV infection induces the accumulation of proteins in neural cells, but the detailed mechanisms underlying the accumulation and how it triggers neurological diseases remain unclear. There are also no established methods to specifically treat viral encephalitis, which can be caused by various types of viruses.
In the current study published in PLOS Pathogens, the research team including Shintaro Kobayashi and Kentaro Yoshii of Hokkaido University focused on autophagy to clarify how protein aggregates form in cells after WNV infection.
The researchers first identified the viral protein, called capsid protein, that induces accumulation of proteins in neural cells by having viral encoded-proteins expressed in cultured neural cells. The capsid protein induced accumulation and aggregation of proteins in the infected cells by inhibiting autophagy, a cellular digestive system. They also found that the capsid protein does so by disrupting an autophagy-inducing factor called AMP-activated protein kinase (AMPK). When they treated the infected cells with a drug that induces autophagy, protein aggregation and cell death were both suppressed.
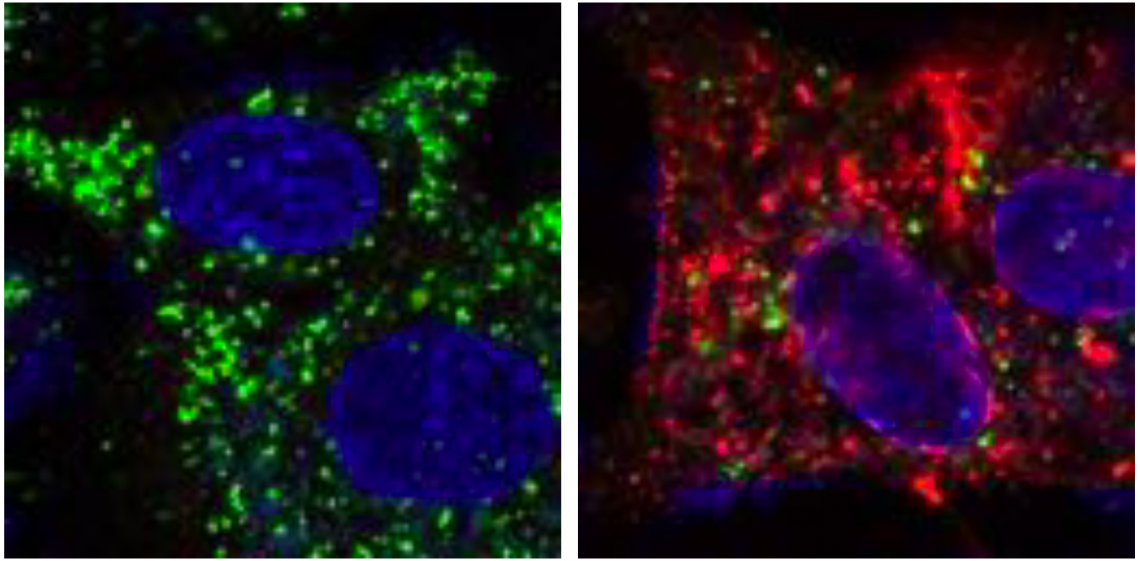
An autophagy related protein LC3, seen in normal cells (left, green) were significantly reduced in WNV infected cells (right). The red signals (E protein) show the infection of WNV. (Kobayashi S. and Yoshii K., et al, PLOS Pathogens. January 24, 2020)
Furthermore, a study using a mouse model demonstrated that WNV with mutations in the capsid protein was unable to harm neural cells or cause encephalitis. These findings suggest WNV inhibits autophagy through the capsid protein and the resulting accumulation of protein is involved in the onset of central nervous system disorders.
“Autophagy anomalies are involved in triggering various diseases, including neurodegenerative illnesses such as Alzheimer’s disease. So, our finding could help elucidate the pathology of West Nile fever as well as various diseases associated with autophagy anomalies, and to develop treatment methods,” said Shintaro Kobayashi of the research team at Hokkaido University.
Funding information:
The study was supported by the Japan Society for the Promotion of Science (JSPS) KAKENHI (15K19069, 24880002, 26660220, 15J00686, 16J00854, 16K15032, 17H03910), Japan Agency for Medical Research and Development (AMED)’s Research Program on Emerging and Re-Emerging Infectious Diseases (JK18fk0108036h0002, 19fk0108036h0003), Takeda Science Foundation, and Czech Academy of Sciences.
Contacts:
Assistant Professor Shintaro Kobayashi
Faculty of Veterinary Medicine
Hokkaido University
Email: shin-kobayashi[at]vetmed.hokudai.ac.jp
Associate Professor Kentaro Yoshii
Faculty of Veterinary Medicine
Hokkaido University
Email: kyoshii[at]vetmed.hokudai.ac.jp
Naoki Namba (Media Officer)
Institute for International Collaboration
Hokkaido University
Tel: +81-11-706-2185
Email: en-press[at]general.hokudai.ac.jp