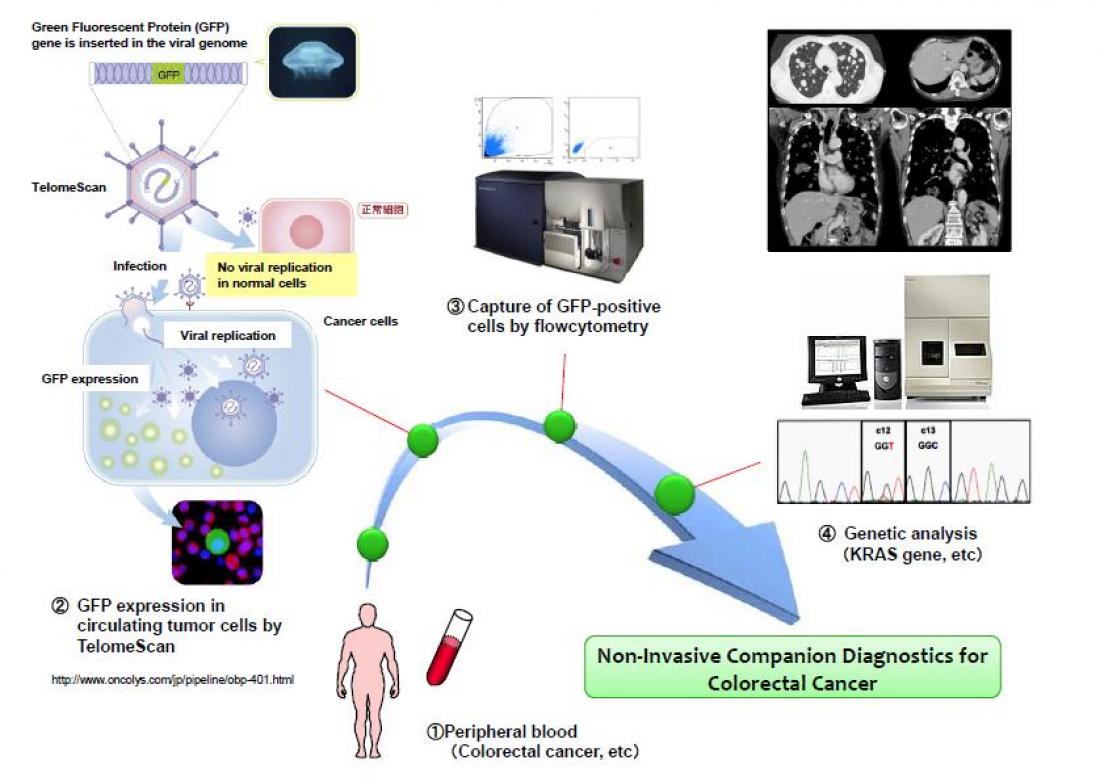
(Okayama, Japan, 10 July 2014) Okayama University medical researchers seek partners to commercialize their clinically proven non-invasive fluorescence virus-guided capture system of human colorectal circulating tumor cells (CTCs) from blood samples for genetic testing. This form of non-invasive companion diagnostics is important for personalized targeted cancer therapy.
The research is led by Professor Toshiyoshi Fujiwara, Okayama University Graduate School of Medicine, Dentistry, and Pharmaceutical Sciences and was published in Gut May 2014 [1]
The key factor in capturing extremely low quantities of live CTCs from millions of background blood leukocytes is targeting the high telomerase activity of malignant tumor cells with green fluorescent protein (GFP) expressing telomerase specific replication adenovirus (OBP-401, TelomeScan [2,3]).
Notably, Professor Fujiwara and colleagues have previously reported on clinical tests on OBP-401 based GFP labelling to detect live CTCs in gasterointestinal and ovarian cancers.
Here, Professor Fujiwara and his team show that their OBP-401 based CTC capture system enables the monitoring of genetic mutations in both epithelial and mesenchymal types of CTCs, thereby opening up the possibility of a new non-invasive companion diagnostic method for genetic testing and personalized medicine.
“This ‘liquid biopsy’ via a simple blood test could be carried out in real time and enables optimized and timely decisions for therapeutic intervention,” state the researchers.
BACKGROUND AND RELEVANCE OF THIS RESEARCH
In 1869 Thomas Ashworth first reported the presence of circulating tumor cells (CTCs) in patients with advanced cancer in his paper entitled: "A case of cancer in which cells similar to those in the tumors were seen in the blood after death". (Australian Medical Journal 14: 146–7, (1869)).
However, it is challenging to detect CTCs because there are very small quantities of CTCs in the bloodstream. The so-called CellSearch system is widely used to detect CTCs, where antibodies are used to target the major epithelial cell surface marker known as the epithelial cell adhesion molecule (EpCAM).
Importantly, recent research has shown the existence of heterogeneous CTCs that have both epithelial and mesenchymal characteristics. This discovery has led to demand for the development of CTC capture systems that are able to detect CTCs irrespective of the epithelial cell marker for cancer treatment with molecularly targeted drugs.
Another important fact is that currently, targeted cancer treatment for individual patients is carried out by analysis of primary tumors. The difficulties with this approach are that the primary tumors contain very few cells that cause metastasis or reoccurrence, and samples for analysis are obtained by needle core biopsies or surgical removal of tumor tissue—which are invasive procedures and prohibit the extraction of tissue from locations inaccessible by surgery.
So there is the need for non-invasive methods capable of detecting CTCs independent of the epithelial cell marker.
DETAILS AND CONCLUSIONS DESCRIBED IN GUT [1]
(1) The OBP-401-based CTC capture system developed by Okayama University can be used to genetic testing of CTCs with mesenchymal characteristics, including GISTs and osteosarcomas.
(2) Combining OBP-401-based CTC capture system and genetic testing enabled the detection of KRAS and BRAF mutations in blood samples taken from patients with colorectal cancer. Notably, these mutations were determined to be identical to the ones seen in the primary tumours of the patients.
(3) This technology requires large scale testing.
For more information on the research, contact
Professor Toshiyoshi Fujiwara,
Department of Gastroenterological Surgery
Okayama University Graduate School of Medicine, Dentistry and Pharmaceutical Sciences
2-5-1 Shikata-cho, Kita-ku
Okayama 700-8558, Japan
EMAIL: [email protected]
References
[1] Shigeyasu K, Tazawa H, Hashimoto Y,et al., Fluorescence virus-guided capturing system of human colorectal circulating tumour cells for non- invasive companion diagnostics, Gut Published Online First: [28 May 2014] doi:10.1136/gutjnl-2014-306957
[2] The manufacturer of OBP-401 (TelomeScan) is Oncolys BioPharma Inc:
http://www.oncolys.com/en/pipeline/obp-1101.html
[3] Patents owned by Oncolys BioPharma Inc:
http://www.oncolys.com/en/company-information/patent.html
Further information on Okayama University
Okayama University
1-1-1 Tsushima-naka , Kita-ku ,
Okayama 700-8530, Japan
Planning and Public Information Division, Okayama University
E-mail: [email protected]
Website: http://www.okayama-u.ac.jp/index_e.html
About Okayama University
Okayama University is one of the largest comprehensive universities in Japan with roots going back to the Medical Training Place sponsored by the Lord of Okayama and established in 1870. Now with 1,300 faculty and 14,000 students, the University offers courses in specialties ranging from medicine and pharmacy to humanities and physical sciences. Okayama University is located in the heart of Japan approximately 3 hours west of Tokyo by Shinkansen.