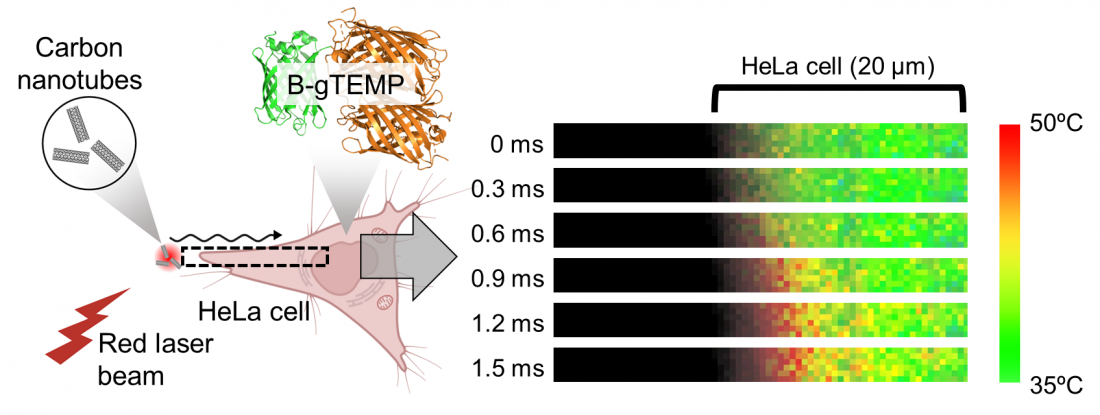
Rapid heat transport in cells. Heat was generated by irradiating carbon nanotubes with a focused red laser beam; the heat then diffused into the adjacent HeLa cell. This process was captured in real time by kilohertz temperature imaging with B-gTEMP.
Researchers from Osaka University and collaborating partners have measured temperature gradients within cells at unprecedented precision. This development will help test long-contentious medical hypotheses and inspire drug development.
Osaka, Japan – Everyone has their temperature measured at a physician's office as a basic indicator of health. Intracellular temperature is also a basic indicator of cellular health; cancer cells are more metabolically active—and thus can have a slightly higher temperature—than healthy cells. However, until now the available tools for testing such hypotheses haven’t been up to the task. In a study recently published in Nano Letters, researchers from Osaka University and collaborating partners have experimentally measured temperature gradients within human cells and at unprecedented precision. This study will open up new directions in drug discovery and medical research.
Many researchers have suspected that transient intracellular temperature gradients have a broader effect on human health than commonly appreciated but were unable to test their hypotheses owing to the limitations of the technology available to them. "Current intracellular thermal detection technology has insufficient spatial, temporal, and readout resolution to answer some long-standing medical hypotheses," explains Kai Lu, lead author, "but our research changes this. Our genetically encoded fluorescent nanothermometer overcomes prior technical hurdles and will be invaluable for testing such hypotheses."
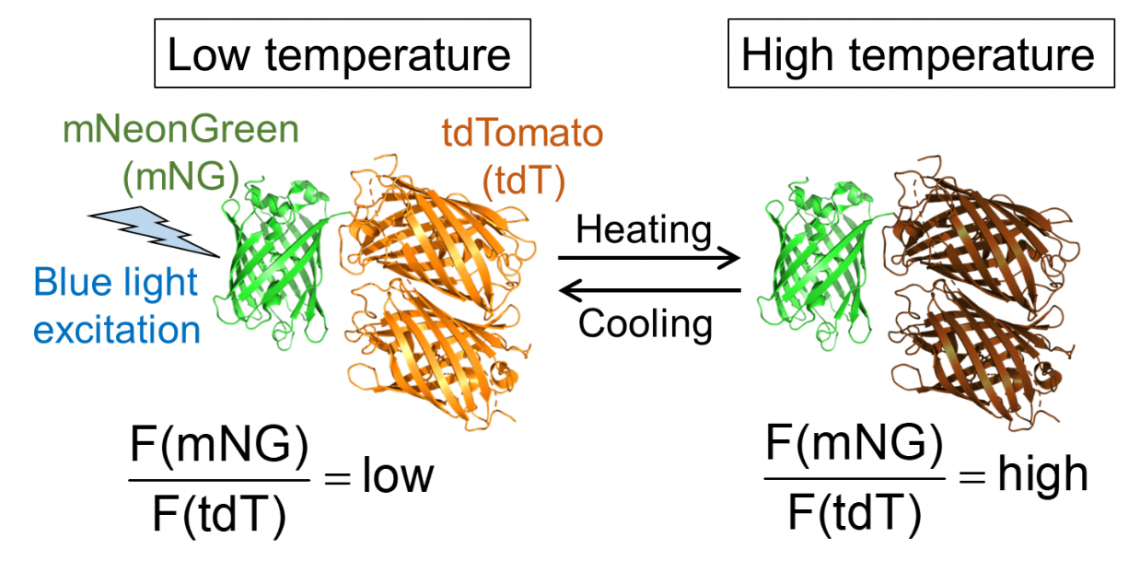
Fig. 1. Molecular design of B-gTEMP and the expected fluorescence response to temperature. F(mNG) and F(tdT) are fluorescence intensity of mNeonGreen and tdTomato, respectively.
The researchers' protein-based nanothermometer is based on modulated fluorescence output that's sensitive to small changes in temperature within cells. Its readout speed is at least 39 times faster than comparable technology, and a thousand times faster than a typical blink of your eye. The nanothermometer enabled the researchers to discover that intracellular heat diffusion is more than 5 times slower than heat diffusion in water. It also showed that the readout resolution is only 0.042°C at physiological temperature, which is an even higher resolution than that in a comparable setup that's several thousand times slower.
"We tested the hypothesis that there's a substantial temperature difference between the cell nucleus and cytoplasm," says Takeharu Nagai, senior author. "We didn't find a significant difference, but test conditions that more closely mimic typical physiology might give different results."
There are several means of improving the functionality of the researchers' nanothermometer. One is to improve how long it lasts under microscopic illumination. Another is to reengineer it to be sensitive to red or infrared light, and thus be less damaging to cells for long-term imaging. In the meantime, researchers now have the technology to realistically probe intracellular temperature gradients, and uncover the physiology that underpins these gradients. Perhaps with this knowledge, drugs can one day be designed to take advantage of this underappreciated aspect of cell physiology.
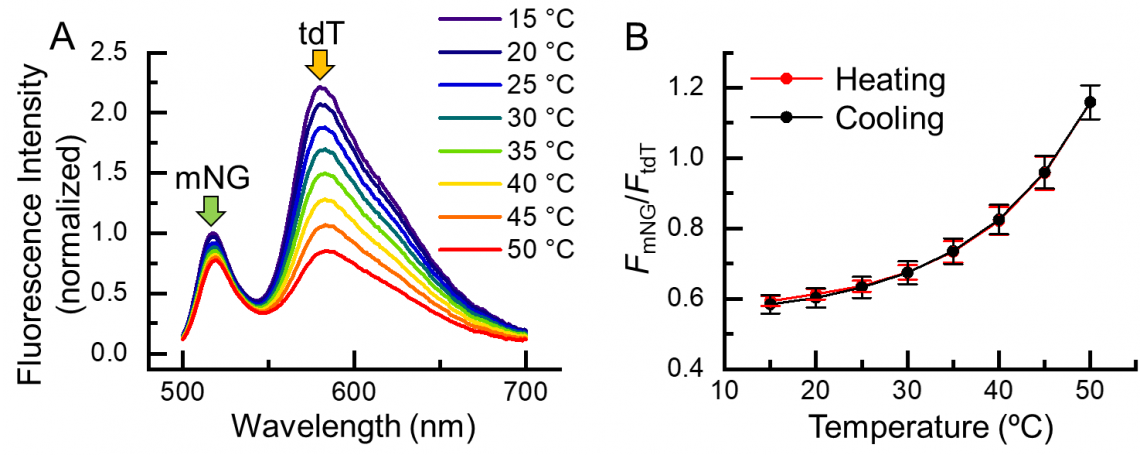
Fig. 2. Temperature response of B-gTEMP. (A) Fluorescence spectrum of B-gTEMP at various temperatures. mNG: mNeonGreen; tdT: tdTomato. (B) Fluorescence intensity ratio of mNG to tdT in response to temperature during a cycle of heating and cooling.
###
The article, "Intracellular heat transfer and thermal property revealed by kilohertz temperature imaging with a genetically encoded nanothermometer," was published in Nano Letters at DOI: https://pubs.acs.org/doi/10.1021/acs.nanolett.2c00608
About Osaka University
Osaka University was founded in 1931 as one of the seven imperial universities of Japan and is now one of Japan's leading comprehensive universities with a broad disciplinary spectrum. This strength is coupled with a singular drive for innovation that extends throughout the scientific process, from fundamental research to the creation of applied technology with positive economic impacts. Its commitment to innovation has been recognized in Japan and around the world, being named Japan's most innovative university in 2015 (Reuters 2015 Top 100) and one of the most innovative institutions in the world in 2017 (Innovative Universities and the Nature Index Innovation 2017). Now, Osaka University is leveraging its role as a Designated National University Corporation selected by the Ministry of Education, Culture, Sports, Science and Technology to contribute to innovation for human welfare, sustainable development of society, and social transformation.
Website: https://resou.osaka-u.ac.jp/en