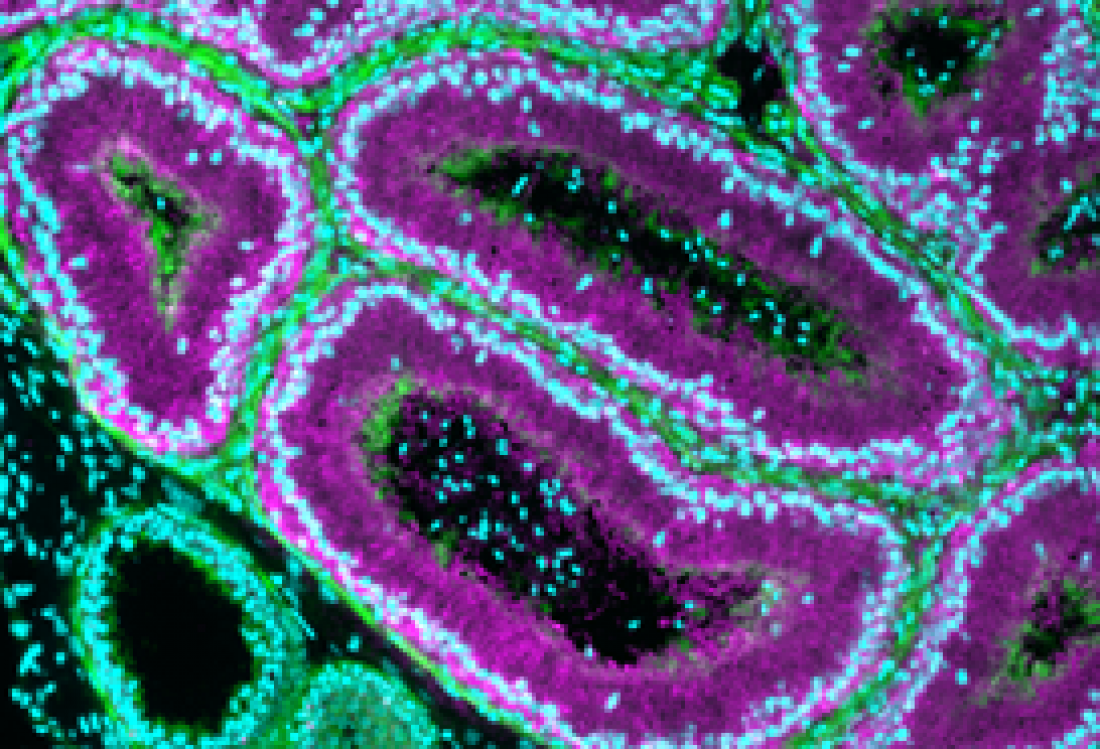
Epididymis (magenta) responding to the protein NICOL coming from the testis. Under the influence of NICOL, spermatozoa, which can be recognized by nuclear staining inside the lumen, become mature and fully fertile.
Researchers from Osaka University reveal a key mechanism underlying sperm maturation; the associated pathway could potentially be manipulated to create safe male contraceptives
Osaka, Japan – Fertility has long been cherished across humankind. Now, in a study published in Nature Communications, researchers from Osaka University have furthered our understanding of sperm maturation. A deeper understanding of this mysterious phenomenon could potentially lead to new male contraception.
In order to create human life, a male and a female sex cell (i.e., gametes) need to fuse together and divide: a process called fertilization. The male gamete, sperm, needs to mature in a tube called the epididymis before it can become a viable cell for fertilization. The mechanisms underlying sperm maturation are relatively understudied and poorly understood.
To better understand the process of sperm maturation, researchers from Osaka University conducted experiments on mice. “The genetic makeup of a mouse can be altered in the lab to study molecular pathways in more detail,” explains senior author Masahito Ikawa. “The advent of genome-editing technology is important; before this technology was available it was challenging to study the mechanisms of sperm maturation.”
The researchers utilized genome-editing technology to manipulate protein expression. Proteins are crucial for cell-to-cell signaling, and are therefore necessary to carry out complex processes like cell maturation. A protein known to be important to sperm maturation is NELL2, a large protein secreted from the testis. NELL2 travels to the epididymis and, similar to how a key interacts with a keyhole, binds to a receptor (ROS1) to signal for sperm maturation to begin.
Crucially, the research team discovered another important protein: ‘NELL2-interacting cofactor for lumicrine signaling’ (NICOL). NICOL forms a tight molecular complex with NELL2 and helps to control the maturation of sperm. Without NICOL, NELL2 is unable to signal properly and sperm maturation is disrupted.
“We found that mice lacking NICOL were sterile, which indicates that NICOL is required for the sperm-maturation pathway to work properly,” says lead author Daiji Kiyozumi. “When NICOL expression was activated, fertility was restored.”
The implications of this study are promising. The development of non-hormonal male contraceptives is one possibility, where NICOL would be targeted to inhibit sperm maturation.
Increasing the expression of NICOL and subsequently recovering sperm maturation could also potentially be used to treat certain cases of infertility.
The next step, however, is to carry out similar experiments in human cells and tissue to see whether findings are replicated.
###
“A small secreted protein NICOL regulates lumicrine-mediated sperm maturation and male fertility” was published in Nature Communications at https://doi.org/10.1038/s41467-023-37984-x
About Osaka University
Osaka University was founded in 1931 as one of the seven imperial universities of Japan and is now one of Japan's leading comprehensive universities with a broad disciplinary spectrum. This strength is coupled with a singular drive for innovation that extends throughout the scientific process, from fundamental research to the creation of applied technology with positive economic impacts. Its commitment to innovation has been recognized in Japan and around the world, being named Japan's most innovative university in 2015 (Reuters 2015 Top 100) and one of the most innovative institutions in the world in 2017 (Innovative Universities and the Nature Index Innovation 2017). Now, Osaka University is leveraging its role as a Designated National University Corporation selected by the Ministry of Education, Culture, Sports, Science and Technology to contribute to innovation for human welfare, sustainable development of society, and social transformation.
Website: https://resou.osaka-u.ac.jp/en