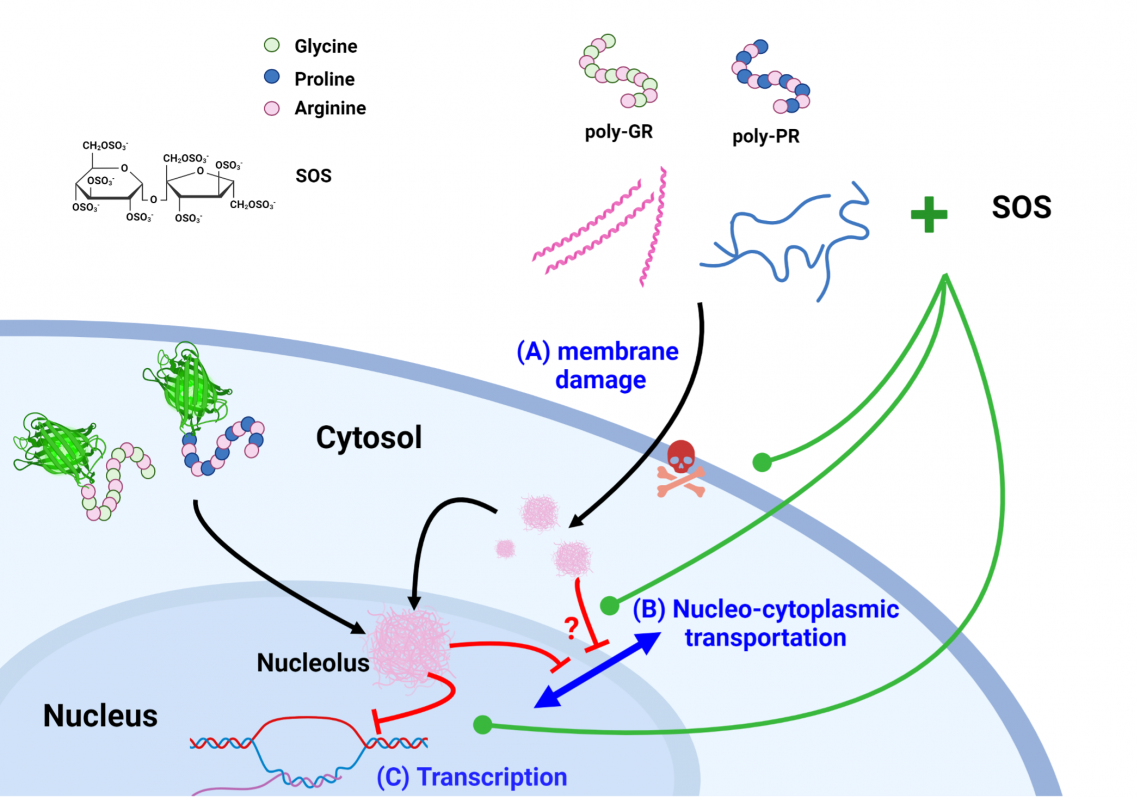
Schematic representation of mechanisms involved in poly-GR/-PR-induced neuronal degradation and the potential therapeutic action of the sulfated disaccharide SOS.
Amyotrophic Lateral Sclerosis (ALS) and Frontotemporal Dementia (FTD) are neurodegenerative diseases that commonly occur in middle-aged people. FTD is second only to Alzheimer's disease in terms of dementia prevalence. Both ALS and FTD arise from neuronal degeneration through mechanisms that remain unclear. Dr. Yun-Ru (Ruby) Chen's team in the Genomics Research Center (GRC), Academia Sinica recently discovered a new pathological mechanism for neuronal degeneration using synthetic peptides1. They also discovered that a disaccharide can increase neuronal survival and reduce degeneration. The result provides therapeutic strategies for future treatment of these neurodegenerative diseases. The study was published in the top international journal "Science Advances" on February 23, 2024.
ALS patients experience progressive muscle atrophy and paralysis due to the degeneration of motor neurons in the spinal cord, brainstem, or motor cortex. Renowned scientist Stephen Hawking and former American baseball star Lou Gehrig had ALS. On the other hand, FTD patients exhibit symptoms such as behavioral deterioration and language impairments due to the degeneration of neurons in the frontal and temporal lobes. Hollywood action star Bruce Willis announced his retirement in 2022 due to FTD.
Dr. Chen's laboratory has long been dedicated to studying neurodegenerative diseases related to protein misfolding. ALS and FTD differ in clinical symptoms, but they share many pathological features and genetic variations. Clinical data shows that >90% of ALS and ~70% of FTD patients are sporadic cases, making it difficult to analyze the underlying genetic variations related to disease. However, in cases found in the same family, the most common genetic mutation for both diseases originates from expansion of the GGGGCC (G4C2) sequence2 in the C9ORF72 gene.
First discovery of poly-GR/-PR interfering nucleic acid mechanisms in cells
The research team first used synthetic poly-GR/-PR peptides with different repeats. Experiments confirmed that repeats of poly-GR for more than25–30 times reduced neuron survival and damaged cell and nuclear membranes. The study also found that the damage occurred through the interaction with nucleic acids, impairing important cellular functions such as DNA, RNA and protein synthesis. Further experiments using cells expressing poly-GR/-PR provided the same results, thus verifying this disease mechanism for the first time. The finding elucidates the pathological mechanism of the G4C2 repeat number in patients.
In collaboration with Dr. U-Ser Jeng's team at the National Synchrotron Radiation Research Center (NSRRC), the team further examined the structures of poly-GR and poly-PR using Small-Angle X-ray Scattering (SAXS). Together with computer simulations, the SAXS data showed that when poly-GR repeats exceed 25–30 times, soft helical structures can form. These results correlated the toxicity of soft helical structures as a function of the repeat length of poly-GR and poly-PR.
Sulfated disaccharide neutralizes the toxicity from poly-GR/PR
Because mature neurons cannot proliferate, increasing cell viability is the goal of therapeutics for neurodegenerative diseases such as ALS and FTD. To identify possible therapeutics, the research team used a library of heparan sulfate molecules established in the laboratory of Dr. Shang-Cheng Hung, Distinguished Research Fellow in GRC. From the screening, a sulfated disaccharide (sucrose octasulfate, SOS) showed the highest potency in reducing degradation of neurons from neuroblastoma and ALS iPSC-derived cells.
The team further found SOS can neutralize poly-GR/PR-induced neuronal degradation and demonstrated an improvement of motor behavior in Drosophila and mouse models. The result provides a possible future therapeutic strategy for ALS and FTD.
The study was led by Dr. Yun-Ru (Ruby) Chen and performed mainly by Ph.D. student Yu-Jen Chang of the Taiwan International Graduate Program - Interdisciplinary Neuroscience (TIGP-INS) in collaboration with Drs. U-Ser Jeng, Shang-Cheng Hung, Chi-Kuang Yao (Institute of Biological Chemistry, AS), Hung-Chih Kuo (Institute of Cellular and Organismic Biology, AS), and Yi-Chung Lee from the Department of Neurology at Taipei Veterans General Hospital.
A full study “Sulfated Disaccharide Protects Membrane and DNA Damages from Arginine-rich Dipeptide Repeats in ALS” was published in Science Advances and can be read online at: https://www.science.org/doi/10.1126/sciadv.adj0347
- poly-GR and poly-PR dipeptide repeats.
- The G4C2 repeat number of C9ORF72 gene in healthy individuals is about 2 to 24, however, the repeats in ALS/FTD patients may expand from a few hundred to even thousands of times. When G4C2 repeats are highly expanded, they can trigger non-ATG-initiated translation, leading to the production of five dipeptide repeats, including poly-GA, poly-GR, poly-GP, poly-PR, and poly-PA, which form inclusion bodies in cells after misfolding. Clinically, these proteinaceous inclusions have been found in various regions in the nervous system, including the hippocampus, frontal lobe, motor cortex, spinal cord, and cerebellum, with poly-GA being the most abundant one. Among them, the hydrophilic poly-GR and poly-PR possess the highest toxicity, distinct from the toxicity mechanism of amyloid deposits.