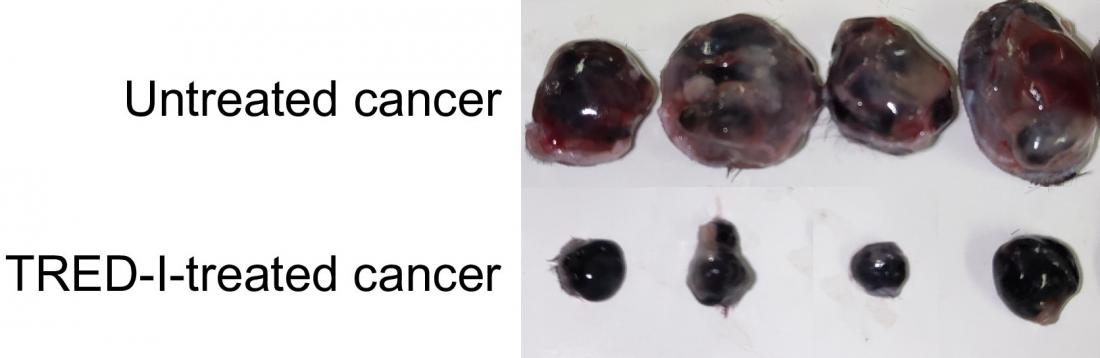
Compared with untreated cancer, the TRED-I system significantly reduced cancer size in mice models. (Xin Sun, et al. Proceedings of the National Academy of Sciences. January 29, 2024)
Joint release by Hokkaido University, Toyo University and University of Missouri.
Major histocompatibility complex (MHC) class I molecules are an immune complex present on the surface of all cells in humans. MHC class I molecules are a prerequisite for the immune system to recognize and eliminate cancer. When cancer cells are faced with pressure from the immune system, they actively reduce their MHC class I molecules, so cancer cells can hide from drawing the attention of CD8+ T cells, the immune system's primary cancer-fighting cells.
Researchers in Japan and the United States, led by Professor Koichi Kobayashi of Hokkaido University and Texas A&M Health Center, and Dr. Paul de Figueiredo, Bond LSC principal investigator and NEXTGEN Precision Health endowed professor at the University of Missouri, have developed technology to robustly augment the amount of MHC class I in cancer cells. This development, a novel method for boosting the immune system's capability to detect and eliminate cancer cells, was published in the journal Proceedings of the National Academy of Sciences.
“Our discovery has the potential to transform the way we approach cancer treatment.” says Kobayashi. “Our technology enables us to specifically target immune responsive genes and activate the immune system against cancer cells, offering hope to those who are resistant to current immunotherapy.”
Kobayashi and his team previously identified a gene, called NLRC5, that regulates MHC class I levels. They further found that NLRC5 is suppressed by turning off molecular switches existing on DNA in cancers—through a process called DNA methylation—to reduce levels of MHC class I.
Their technology, known as TRED-I (Targeted Reactivation and Demethylation for MHC-I) system, was able to restore DNA methylation of NLRC5 gene and further activate NLRC5, thus increasing MHC class I levels in cancer without causing severe side effects.
“New modalities for fighting cancer like this are desperately needed because we have few solutions to fight some cancer types,” said de Figueiredo. “This is a radically new approach, and I’ve felt lucky to be part of it.”
TRED-I was tested with animal cancer models. It reduced tumor sizes significantly and increased activity of cytotoxic CD8+ T cells. When used in conjunction with existing immunotherapy, TRED-I markedly enhanced treatment efficacy.
Unexpectedly, the TRED-I system was effective for the tumor distantly located from the original targeted tumor, showing potential to treat metastasized cancers.
“This work is the culmination of our team’s research over the past decade,” Kobayashi concludes. “It’s great to shed light on moving our findings to potential clinical applications. We believe with further refinement, the TRED-I system could contribute significantly to cancer therapy.”
Further research will focus on enabling direct delivery of the TRED-I system in cancer patients. Such drugs could improve the efficacy of the immune system in eliminating cancer and would be also able to improve the response to existing therapy.